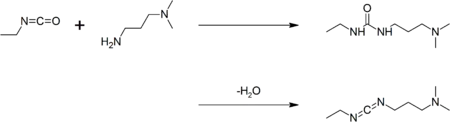1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide
1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC, EDAC or EDCI) is a water-soluble carbodiimide usually handled as the hydrochloride.[1] It is typically employed in the 4.0-6.0 pH range. It is generally used as a carboxyl activating agent for the coupling of primary amines to yield amide bonds. Additionally, EDC can also be used to activate phosphate groups in order to form phosphomonoesters and phosphodiesters. Common uses for this carbodiimide include peptide synthesis, protein crosslinking to nucleic acids, but also in the preparation of immunoconjugates. EDC is often used in combination with N-hydroxysuccinimide (NHS) for the immobilisation of large biomolecules. Recent work has also used EDC to assess the structure state of uracil nucleobases in RNA.[2][3]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
3-(Ethyliminomethyleneamino)-N,N-dimethylpropan-1-amine | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.015.982 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H17N3 | |
| Molar mass | 155.245 g·mol−1 |
| Hazards | |
| Safety data sheet | External MSDS (HCl Salt) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
EDC is commercially available. It may be prepared by coupling ethyl isocyanate to N,N-dimethylpropane-1,3-diamine to give a urea, followed by dehydration:[4]
Mechanism
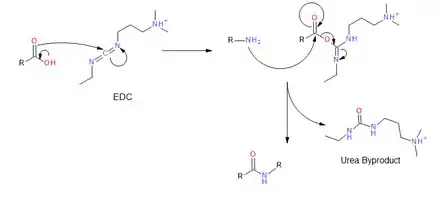
EDC couples primary amines to carboxylic acids by creating an activated ester leaving group. First, the carbonyl of the acid attacks the carbodiimide of EDC, and there is a subsequent proton transfer. The primary amine then attacks the carbonyl carbon of the acid which forms a tetrahedral intermediate before collapsing and discharging the urea byproduct. The desired amide is obtained.[5]
References
- Richard S. Pottorf, Peter Szeto (2001). "1-Ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride". e-EROS Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.re062.
- Mitchell, D; Renda, A; Douds, C; Babitzke, P; Assmann, S; Bevilacqua, P (2019). "In vivo RNA structural probing of uracil and guanine base-pairing by 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC)". RNA. 25 (1): 147-157. doi:10.1261/rna.067868.118. PMC 6298566. PMID 30341176.
- Wang, PY; Sexton, AN; Culligan, WJ; Simon, MD (2019). "Carbodiimide reagents for the chemical probing of RNA structure in cells". RNA. 25 (1): 135–146. doi:10.1261/rna.067561.118. PMC 6298570. PMID 30389828.
- Sheehan, John; Cruickshank, Philip; Boshart, Gregory (1961). "A Convenient Synthesis of Water-Soluble Carbodiimides". J. Org. Chem. 26 (7): 2525. doi:10.1021/jo01351a600.
- "Carbodiimide Crosslinker Chemistry - US". www.thermofisher.com. Retrieved 2019-05-10.
Further reading
- López-Alonso, JP; Diez-Garcia, F; Font, J; Ribó, M; Vilanova, M; Scholtz, JM; González, C; Vottariello, F; Gotte, G; Libonati, M; Laurents, DV (2009). "Carbodiimide EDC Induces Cross-Links That Stabilize RNase A C-dimer against Dissociation: EDC Adducts Can Affect Protein Net Charge, Conformation and Activity". Bioconjugate Chemistry. 20 (8): 1459. doi:10.1021/bc9001486.
- Nakajima, N; Ikada, Y (1995). "Mechanism of Amide Formation by Carbodiimide for Bioconjugation in Aqueous Media". Bioconjugate Chemistry. 6: 123. doi:10.1021/bc00031a015.
External links
- EDCI coupling - Synthetic protocols from organic-reaction.com