Dienone-phenol rearrangement
The dienone-phenol rearrangement is a reaction in organic chemistry first reported in 1921 by Auwers and Ziegler. A common example of dienone-phenol rearrangement is 4,4-disubstituted cyclohexadienone converting into a stable 3,4-disubstituted phenol in presence of acid. A similar rearrangement is possible with 2,2-disubstituted cyclohexadienone to its corresponding disubstituted phenol. Usually this type rearrangement is a spontaneous unless the presence of a dichloromethyl group at 3rd position or 4th position is blocked with any non hydrogen groups.[1]


Reaction mechanism
The reaction mechanism of 4,4-disubstituted cyclohexadienones to 3,4-disubstituted phenol is illustrated here.
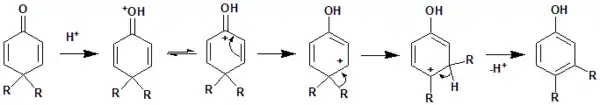
The migration tendency for the two different groups (R) present at either 4,4 position or 2,2 position can be determined by comparing the relative stability of intermediate Carbocation formed in the time of rearrangement. In case of acid-promoted conditions, the migration tendencies few groups are like this COOEt > Phenyl (or alkyl);[2] Phenyl > Methyl;[3] vinyl > Methyl;[4] Methyl > alkoxy and alkoxy > Phenyl.[5] In some cases such as allyl and benzyl group, the actual rearrangement might undergo through the Cope Rearrangement.[6] [7]Apart from acid catalysis, the dienone-phenol rearrangement is also possible in presence of base. The dienone-phenol rearrangement found helpful in various synthetic protocols use towards the synthesis of steroids,[8] anthracene,[9] phenanthrene,[10] etc.
References
- Comprehensive Organic Name Reactions and Reagents - "Dienone‐Phenol Rearrangement". John Wiley & Sons. 15 September 2010. p. 900. ISBN 9780470638859.
- "Molecular rearrangements with ethoxycarbonyl group migrations. 1. The rearrangement of epoxides". J. Am. Chem. Soc. 98 (15): 4581. July 1, 1976. doi:10.1021/ja00431a042.
- Arnold, Richard T. (May 1, 1949). "The Dienone-Phenol Rearrangement. II. Rearrangement of 1-Keto-4-methyl-4-phenyl-1,4-dihydronaphthalene". J. Am. Chem. Soc. 71 (5): 1781. doi:10.1021/ja01173a071.
- "Acid-Catalyzed Migration of the Vinyl Substituent in the Dienone-Phenol Rearrangement". J. Org. Chem. 53 (12): 2866. November 4, 1987. doi:10.1021/jo00247a046.
- "Dienone-Phenol Rearrangement of 2-Phenylo-quinolacetate1". J. Am. Chem. Soc. 71 (5): 1781. May 1, 1949. doi:10.1021/ja01173a071.
- "Acid-Catalyzed Rearrangements of Cyclohexa-2,4-dien-l-ones. Competitive [1,2] and [1,5] Migrations". Journal of the American Chemical Society. 92 (2): 432. 1970.
- Bernard, Miller (1965). "Dienone-Phenol Rearrangements of 6-Allylcyclohexa-2,4-dienones and 4-Allylcyclohexa-2,5-dienones. Acid Catalysis of Cope Rearrangements1". Journal of the American Chemical Society. 87 (22): 5115.
- "The Preparation and Dienone-Phenol Rearrangement of 2-Bromo-1,4-androstadien-17-ol-3-one 17-Hexahydrobenzoate". J. Am. Chem. Soc. 70 (5): 1911. 1948. doi:10.1021/ja01185a075.
- "The Rearrangement of Some Disubstituted Cyclic Ketones: An Unusual Dienone—Phenol Rearrangement". J. Am. Chem. Soc. 74 (5): 1259. March 1, 1952. doi:10.1021/ja01125a035.
- "The Dienone-Phenol Rearrangement. III. Rearrangement of 1 -Keto-4,4-tetramethylene-1,4-dihydronaphthalene". Journal of the American Chemical Society. 72 (7): 3153. 1950.