Diethyl tartrate
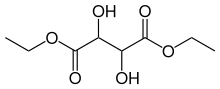
Diethyl tartrate is an organic compound with the formula (HOCHCO2Et)2 (Et = ethyl). Three stereoisomers exist, R,R-, S,S-, and R,S (=S,R-). They are the ethyl esters of the respective R,R-, S,S-, and R,S-tartaric acids. The R,R- and S,S- isomers are enantiomeric, being mirror images. The meso stereoisomer is not chiral. The chiral isomer is far more common.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Diethyl 2,3-dihydroxybutanedioate | |
| Other names
Diethyl 2,3-dihydroxysuccinate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C8H14O6 | |
| Molar mass | 206.194 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.204 g/mL |
| Boiling point | 280 °C (536 °F; 553 K) |
| low | |
| -113.4·10−6 cm3/mol | |
| Hazards | |
| Safety data sheet | Diethyl D-Tartrate MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
In the Sharpless epoxidation, diethyl tartrate and titanium isopropoxide form a chiral catalyst in situ.[1]
References
- Hill, J. Gordon; Sharpless, K. Barry; Exon, Christopher M.; Regenye, Ronald (1985). "Enantioselective Epoxidation of Allylic Alcohols: (2S,3S)-3-Propyloxiranemethanol". Organic Syntheses. 63: 66. doi:10.15227/orgsyn.063.0066.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.