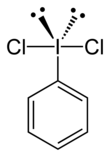
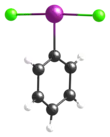
Iodobenzene dichloride
Iodobenzene dichloride (PhICl2) is a complex of iodobenzene with chlorine. As a reagent for organic chemistry, it is used as an oxidant and chlorinating agent.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
(Dichloro-λ3-iodanyl)benzene | |||
| Other names
Iodosobenzene dichloride; Phenyliodine(III) dichloride; Phenyliodo dichloride; Phenyliodoso chloride; Phenylchloroiodonium chloride; Dichloroiodobenzene; Iododichlorobenzene | |||
| Identifiers | |||
3D model (JSmol) |
|||
| Abbreviations | IBD | ||
| ChemSpider | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| Properties | |||
| C6H5Cl2I | |||
| Molar mass | 274.91 g·mol−1 | ||
| Appearance | Yellow solid | ||
| Density | 2.2 g/cm3 | ||
| Melting point | 115 to 120 °C (239 to 248 °F; 388 to 393 K) (decomposes) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Chemical structure
Single-crystal X-ray crystallography has been used to determine its structure; as can be predicted by VSEPR theory, it adopts a T-shaped geometry about the central iodine atom.[2][3]
Preparation
Iodobenzene dichloride is not stable, and is not commonly available commercially. It is prepared by passing chlorine gas through a solution of iodobenzene in chloroform, from which it precipitates.[4] The same reaction has been reported at pilot plant scale (20 kg) as well.[5]
- Ph-I + Cl2 → PhICl2
An alternate preparation involving the use of chlorine generated in situ by the action of sodium hypochlorite on hydrochloric acid has also been described.[6]
Reactions
Iodobenzene dichloride is hydrolyzed by basic solutions to give iodosobenzene (PhIO),[7] and is oxidized by sodium hypochlorite to give iodoxybenzene (PhIO2).[8]
In organic synthesis, iodobenzene dichloride is used as a reagent for the selective chlorination of alkenes[1] and alkynes.[9]
References
- Phenyliodine(III) Dichloride, David W. Knight and Glen A. Russell, in Encyclopedia of Reagents for Organic Synthesis, 2001, John Wiley & Sons, Ltd doi:10.1002/047084289X.rp071
- E. M. Archer and T. G. van Schalkwy (1953). "The crystal structure of benzene iododichloride". Acta Crystallogr. 6: 88–92. doi:10.1107/S0365110X53000193.
- J. V. Carey; P. A. Chaloner; P. B. Hitchcock; T. Neugebauer; K. R. Seddon (1996). "Synthesis and decomposition of dichloroiodoarenes - An improved low temperature X-ray structure of dichloroiodobenzene and the structure OFORO-2,3,5,6-TETRAKIS(CHLOROMETHYL)-4-METHYLBENZENE". J. Chem. Res. 358 (8): 2031–.
- H. J. Lucas and E. R. Kennedy. "Iodobenzene dichloride". Organic Syntheses.; Collective Volume, 3, p. 482
- Zanka, Atsuhiko; Takeuchi, Hiroki; Kubota, Ariyoshi (1998). "Large-Scale Preparation of Iodobenzene Dichloride and Efficient Monochlorination of 4-Aminoacetophenone". Organic Process Research & Development. 2 (4): 270. doi:10.1021/op980024e.
- Zhao, Xue-Fei; Zhang, Chi (2007). "Iodobenzene Dichloride as a Stoichiometric Oxidant for the Conversion of Alcohols into Carbonyl Compounds; Two Facile Methods for Its Preparation". Synthesis. 2007 (4): 551. doi:10.1055/s-2007-965889.
- H. J. Lucas, E. R. Kennedy, and M. W. Formo (1955). "Iodosobenzene". Organic Syntheses.CS1 maint: multiple names: authors list (link); Collective Volume, 3, p. 483
- M. W. Formo and John R. Johnson (1955). "Iodoxybenzene: B. Hypochlorite oxidation of iodobenzene dichloride". Organic Syntheses.; Collective Volume, 3, p. 485
- Michael E. Jung and Michael H. Parker (1997). "Synthesis of Several Naturally Occurring Polyhalogenated Monoterpenes of the Halomon Class". Journal of Organic Chemistry. 62 (21): 7094–7095. doi:10.1021/jo971371. PMID 11671809.
Further reading
- Tanner, Dennis D; Van Bostelen, P. B. (1967). "Free-radical chlorination reactions of iodobenzene dichloride". Journal of Organic Chemistry. 32 (5): 1517–1521. doi:10.1021/jo01280a047.