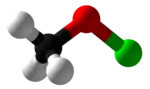
Methyl hypochlorite
Methyl hypochlorite is the simplest of the organic alkyl hypochlorites. It is an unstable compound that can be produced by the reaction of methanol with hypochlorous acid.[1] It was first synthesized by Sandmeyer in the 1880s.[2]
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Methyl hypochlorite | |||
| Other names
(Chlorooxy)methane; Hypochlorous acid methyl ester; Methoxy chloride | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CH3ClO | |||
| Molar mass | 66.48 g·mol−1 | ||
| Appearance | Gas | ||
| Odor | Pungent | ||
| Density | 1.058 g/cm3 | ||
| Melting point | −120.4 °C (−184.7 °F; 152.8 K) | ||
| Boiling point | 9.18 °C (48.52 °F; 282.33 K) | ||
| Decomposes | |||
Refractive index (nD) |
1.343 | ||
| Hazards | |||
| R-phrases (outdated) | R3 R8 R23/24/25 R35 | ||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Methyl hypochlorite forms in the Earth's atmosphere by a reaction between ClO and CH3OO and is thought to be an important species in ozone destruction over the Arctic and Antarctic regions.[3]
See also
- tert-Butyl hypochlorite, a useful and relatively stable organic hypochlorite
References
- Taylor, M. C.; MacMullin, R. B.; Gammal, C. A. (February 1925). "Hypochlorous Acid and the Alkyl Hypochlorite". Journal of the American Chemical Society. 47 (2): 395–403. doi:10.1021/ja01679a017.
- Sandmeyer, Traugott (January 1886). "Ueber Aethyl- und Methylhypochlorit". Berichte der Deutschen Chemischen Gesellschaft. 19 (1): 857–861. doi:10.1002/cber.188601901196.
- Helleis, Frank; Crowley, John; Moortgat, Geert (15 August 1994). "Temperature dependent CH3OCl formation in the reaction between CH3O2 and ClO". Geophysical Research Letters. 21 (17): 1795–1798. doi:10.1029/94GL01280.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.