TST (gene)
Thiosulfate sulfurtransferase is an enzyme that in humans is encoded by the TST gene.[5][6]
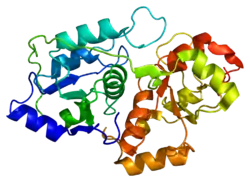
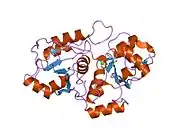
The product of this gene is a mitochondrial matrix enzyme that is encoded by the nucleus. It may play roles in cyanide detoxification, the formation of iron-sulfur proteins, and the modification of sulfur-containing enzymes.
The gene product contains two highly conservative domains (rhodanese homology domains), suggesting these domains have a common evolutionary origin.[6]
References
- GRCh38: Ensembl release 89: ENSG00000128311 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000044986 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Pallini R, Guazzi GC, Cannella C, Cacace MG (Dec 1991). "Cloning and sequence analysis of the human liver rhodanese: comparison with the bovine and chicken enzymes". Biochem Biophys Res Commun. 180 (2): 887–93. doi:10.1016/S0006-291X(05)81148-9. PMID 1953758.
- "Entrez Gene: TST thiosulfate sulfurtransferase (rhodanese)".
Further reading
- Pecci L, Pensa B, Costa M, et al. (1976). "Reaction of rhodanese with dithiothreitol". Biochim. Biophys. Acta. 445 (1): 104–11. doi:10.1016/0005-2744(76)90163-7. PMID 986188.
- Polo CF, Vazquez ES, Caballero F, et al. (1993). "Heme biosynthesis pathway regulation in a model of hepatocarcinogenesis pre-initiation". Comp. Biochem. Physiol., B. 103 (1): 251–6. doi:10.1016/0305-0491(92)90440-3. PMID 1451437.
- Lewis JL, Rhoades CE, Gervasi PG, et al. (1991). "The cyanide-metabolizing enzyme rhodanese in human nasal respiratory mucosa". Toxicol. Appl. Pharmacol. 108 (1): 114–20. doi:10.1016/0041-008X(91)90274-I. PMID 2006499.
- Malliopoulou VA, Rakitzis ET, Malliopoulou TB (1989). "Inactivation of rhodanese from human gastric mucosa and stomach adenocarcinoma by 2,4, 6-trinitrobenzenesulphonate and by 4,4'-diisothiocyanatostilbene-2,2'-disulphonate". Anticancer Res. 9 (4): 1133–6. PMID 2817794.
- Vazquez E, Buzaleh AM, Wider E, Batlle AM (1987). "Red blood cell rhodanese: its possible role in modulating delta-aminolaevulinate synthetase activity in mammals". Int. J. Biochem. 19 (2): 217–9. doi:10.1016/0020-711X(87)90337-5. PMID 3471602.
- Pallini R, Martelli P, Bardelli AM, et al. (1987). "Normal rhodanese activity in leukocytes from Leber patients: enzyme characterization and activity levels". Neurology. 37 (12): 1878–80. doi:10.1212/wnl.37.12.1878. PMID 3479705.
- Pagani S, Galante YM (1983). "Interaction of rhodanese with mitochondrial NADH dehydrogenase". Biochim. Biophys. Acta. 742 (2): 278–84. doi:10.1016/0167-4838(83)90312-6. PMID 6402020.
- Mimori Y, Nakamura S, Kameyama M (1984). "Regional and subcellular distribution of cyanide metabolizing enzymes in the central nervous system". J. Neurochem. 43 (2): 540–5. doi:10.1111/j.1471-4159.1984.tb00932.x. PMID 6588145.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Merrill GA, Butler M, Horowitz PM (1993). "Limited tryptic digestion near the amino terminus of bovine liver rhodanese produces active electrophoretic variants with altered refolding". J. Biol. Chem. 268 (21): 15611–20. PMID 8340386.
- Aita N, Ishii K, Akamatsu Y, et al. (1997). "Cloning and expression of human liver rhodanese cDNA". Biochem. Biophys. Res. Commun. 231 (1): 56–60. doi:10.1006/bbrc.1996.6046. PMID 9070219.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, et al. (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Dunham I, Shimizu N, Roe BA, et al. (1999). "The DNA sequence of human chromosome 22". Nature. 402 (6761): 489–95. doi:10.1038/990031. PMID 10591208.
- Wieprecht T, Apostolov O, Beyermann M, Seelig J (2001). "Interaction of a mitochondrial presequence with lipid membranes: role of helix formation for membrane binding and perturbation". Biochemistry. 39 (50): 15297–305. doi:10.1021/bi001774v. PMID 11112515.
- Picton R, Eggo MC, Merrill GA, et al. (2002). "Mucosal protection against sulphide: importance of the enzyme rhodanese". Gut. 50 (2): 201–5. doi:10.1136/gut.50.2.201. PMC 1773108. PMID 11788560.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Gevaert K, Goethals M, Martens L, et al. (2004). "Exploring proteomes and analyzing protein processing by mass spectrometric identification of sorted N-terminal peptides". Nat. Biotechnol. 21 (5): 566–9. doi:10.1038/nbt810. PMID 12665801.
- Kwiecień I, Sokołowska M, Luchter-Wasylewska E, Włodek L (2004). "Inhibition of the catalytic activity of rhodanese by S-nitrosylation using nitric oxide donors". Int. J. Biochem. Cell Biol. 35 (12): 1645–57. doi:10.1016/S1357-2725(03)00005-0. PMID 12962704.
- Ota T, Suzuki Y, Nishikawa T, et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.