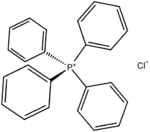
Tetraphenylphosphonium chloride
Tetraphenylphosphonium chloride is the chemical compound with the formula (C6H5)4PCl, abbreviated Ph4PCl or PPh4Cl. Tetraphenylphosphonium and especially tetraphenylarsonium salts were formerly of interest in gravimetric analysis of perchlorate and related oxyanions. This colourless salt is used to generate lipophilic salts from inorganic and organometallic anions. Thus, Ph4P+ is useful as a phase-transfer catalyst, again because it allows inorganic anions to dissolve in organic solvents.
 | |
| Names | |
|---|---|
| IUPAC name
Tetraphenylphosphonium chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.016.265 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C24H20ClP | |
| Molar mass | 374.84 g/mol |
| Appearance | colourless solid |
| Density | 1.27 g dm−3 |
| Melting point | 272 to 274 °C (522 to 525 °F; 545 to 547 K) |
| Hazards | |
| R-phrases (outdated) | 36/37/38 |
| S-phrases (outdated) | 26-36 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure and basic properties
The cation is tetrahedral. PPh4Cl crystallises as the anhydrous salt,[1] which is normal item of commerce, as well as a monohydrate[2] and a dihydrate.[3]
In X-ray crystallography, PPh+
4 salts are of interest as they often crystallise easily. The rigidity of the phenyl groups facilitates packing and elevates the melting point relative to alkyl-based quaternary ammonium salts. Also, since these salts are soluble in organic media, a wide range of solvents can be employed for their crystallisation.
 | 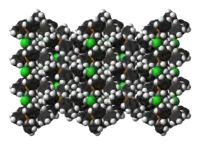 | 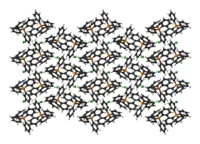 |
of the crystal structure | of the crystal structure |
Preparation
PPh4Cl and many analogous compounds can be prepared by the reaction of chlorobenzene with triphenylphosphine catalysed by nickel salts:[4]
- PhCl + PPh3 → Ph4PCl
The compound was originally prepared as the corresponding bromide salt (CAS No. 2751-90-8), which in turn was synthesized by passing dry oxygen through the reaction of phenylmagnesium bromide and triphenylphosphine.[5] The synthesis probably proceeds via the reaction of the Grignard reagent with triphenylphosphine oxide.
- PhMgBr + Ph3PO → Ph4POMgBr
- Ph4POMgBr + HBr → Ph4PBr + "Mg(OH)Br"
References
- Richardson, J. F.; Ball, J. M.; Boorman, P. M. (1986). "Structure of Tetraphenylphosphonium Chloride". Acta Crystallographica. C42 (9): 1271–1272. doi:10.1107/S0108270186092612.
- Schweizer, E. E.; Baldacchini, C. J.; Rheingold, A. L. (1989). "Tetraphenylphosphonium Chloride Monohydrate, Tetraphenylphosphonium Bromide and Tetraphenylphosphonium Iodide". Acta Crystallographica. C45 (8): 1236–1239. doi:10.1107/S0108270189000363.
- Blake, A. J.; Garner, C. D.; Tunney, J. M. (2003). "Tetraphenylphosphonium Chloride Dihydrate". Acta Crystallographica. E59: o9–o10. doi:10.1107/S1600536802021682.
- Marcoux, David; Charette, André B. (2008). "Nickel-Catalyzed Synthesis of Phosphonium Salts from Aryl Halides and Triphenylphosphine". Adv. Synth. Catal. 350 (18): 2967–2974. doi:10.1002/adsc.200800542.
- Dodonow, J.; Medox, H. (1928). "Zur Kenntnis der Grignardschen Reaktion: Über die Darstellung von Tetraphenyl-phosphoniumsalzen". Berichte der Deutschen Chemischen Gesellschaft. 61 (5): 907–911. doi:10.1002/cber.19280610505.