Thiosulfonate
Thiosulfonates are organosulfur compounds with the formula RSO2SR'. Thiosulfonate esters are produced by oxidation of disulfides or by the reaction or organosulfonyl halides with thiolates.[1]
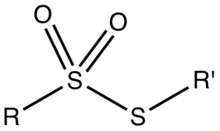
Structure of thiosulfonate ester.
Alkali metal thiosulfonates are the conjugate base of thiosulfuric acid. They are prepared by the reaction of organosulfonyl chlorides with sources of sulfide.[1][2]
See also
- Bunte salts are related organosulfur compounds with the formula RSSO3−
- Thiosulfinate a structurally analogous functional group in a lower oxidation state, with the formula RSS(O)R
References
- Nikolai S. Zefirov, Nikolai V. Zyk, Elena K. Beloglazkina, Andrei G. Kutateladze (1993). "Thiosulfonates: Synthesis, Reactions and Practical Applications". Sulfur Reports. 14: 223–240. doi:10.1080/01961779308055018.CS1 maint: multiple names: authors list (link)
- R. B. Woodward, I. J. Pachter, Monte L. Scheinbaum (1974). "Trimethylene Dithiotosylate And Ethylene Dithiotosylate". Org. Synth. 54: 33. doi:10.15227/orgsyn.054.0033.CS1 maint: uses authors parameter (link)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.