Vagal tone
Vagal tone refers to activity of the vagus nerve, the 10th cranial nerve and a fundamental component of the parasympathetic branch of the autonomic nervous system. This branch of the nervous system is not under conscious control and is largely responsible for the regulation of several body compartments at rest. Vagal activity results in various effects, including: heart rate reduction, vasodilation/constriction of vessels, glandular activity in the heart, lungs, and digestive tract, liver, immune system regulation as well as control of gastrointestinal sensitivity, motility and inflammation.[1]
In this context, tone specifically refers to the continual nature of baseline parasympathetic action that the vagus nerve exerts. While baseline vagal input is constant, the degree of stimulation it exerts is regulated by a balance of inputs from sympathetic and parasympathetic divisions of the autonomic nervous system, with parasympathetic activity generally being dominant. Vagal tone is frequently used to assess heart function, and is also useful in assessing emotional regulation and other processes that alter, or are altered by, changes in parasympathetic activity.[2][3]
Measurements of vagal tone can be performed by means of either invasive or noninvasive procedures. Invasive procedures are in the minority and include vagus nerve stimulation by specific manual, breathing or electrical techniques. Noninvasive techniques mainly rely on the investigation of heart rate and heart rate variability.[4][5][6]
Noninvasive vagal tone quantification
In most cases, vagal tone is not measured directly. Instead the processes affected by the vagus nerve – specifically heart rate and heart rate variability – are measured and used as a surrogate for vagal tone. Increased vagal tone (and thus vagal action) is generally associated with a lower heart rate and increased heart rate variability. However, during graded orthostatic tilt, vagal tone withdrawal is an indirect indicator of cardiovascular fitness.[7]
Vagal innervation of the heart
Heart rate is largely controlled by the heart's internal pacemaker activity. In a healthy heart, the main pacemaker is a collection of cells on the border of the atria and vena cava called the sinoatrial node. Heart cells exhibit automaticity, the ability to generate electrical activity independent of external stimulation. The electrical activity spontaneously generated by the sinoatrial node sets the pace for the rest of the heart.[1]
In absence of external stimuli, sinoatrial pacing generally maintains the heart rate in the range of 60–100 beats per minute (bpm).[8] The two branches of the autonomic nervous system work together to increase or slow the heart rate. The vagus nerve acts on the sinoatrial node, slowing its conduction and modulating vagal tone, via the neurotransmitter acetylcholine and downstream changes to ionic currents and calcium of heart cells.[4] Because of its effect on heart rate, and cardio health, vagal tone can be measured and understood by examining its correlation to heart rate modulation and heart rate variability.[5]
Respiratory sinus arrhythmia
Respiratory sinus arrhythmia (RSA) is typically a benign, normal variation in heart rate that occurs during each breathing cycle: the heart rate increases when breathing in and decreases when breathing out.[1] RSA was first recognized by Carl Ludwig in 1847[9] but is still imperfectly understood.[10] It has been observed in humans from the early stages of life through adulthood,[11][1] and is found in several different species.[12][13][14]
During inhalation, the intra-thoracic pressure lowers due to the contraction and downward movement of the diaphragm and the expansion of the chest cavity. Atrial pressure is also lowered as a result, causing increased blood flow to the heart, which in turn decreases baroreceptors firing response which diminishes vagal tone. This causes an increase in heart rate.[1]
During exhalation, the diaphragm relaxes, moving upward, and decreases the size of the chest cavity, causing an increase in intrathoracic pressure. This increase in pressure inhibits venous return to the heart resulting in both reduced atrial expansion and increased activation of baroreceptors. This relieves the suppression of vagal tone and leads to a decreased heart rate.[1]
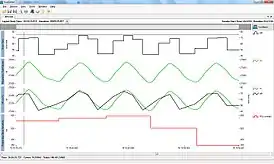

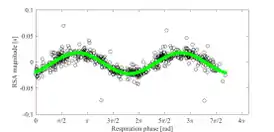
RSA as a vagal tone estimator
Respiratory sinus arrhythmia is frequently used as a noninvasive method for investigating vagal tone, in physiological, behavioral, and several clinical studies.[16][17][18] This can be done using electrocardiography (ECG) recording,[19] although other methods are also being developed that take advantage of the interactions between ECG and respiration.[20][15] Interpretation of RSA measurements must be done with care, however, as several factors including differences between individuals can change the relationship between RSA and vagal tone.[21]
Evolution and physiology
It has been suggested that RSA may have evolved to save energy for both cardiac and respiratory systems by reducing the heart rate[22] and by suppressing ineffective ventilation during the ebb of perfusion (delivery of blood from arteries to capillaries for oxygenation and nutrition).[23][24]
RSA has been found to increase in subjects in resting state and to decrease in states of stress or tension. It is increased in supine position and decreased in prone position, and is on average higher and more pronounced during the day as compared to the night.[22] RSA has also been extensively used to quantify vagal tone withdrawal in graded orthostatic tilt.[7][25]
Typically, expression of RSA decreases with age.[26] However, adults in excellent cardiovascular health, such as endurance runners, swimmers, and cyclists, are likely to have a more pronounced RSA. Professional athletes on average maintain very high vagal tone and consequently higher RSA levels. RSA is less prominent in individuals with diabetes and cardiovascular disease.[27]
Insights into psychology and disease
Vagal tone research has the potential to offer insight into social behavior, social interactions, and human psychology. Much of this work has been focused on newborns and children.[26] Baseline vagal tone can be used either as a potential predictor of behavior or as a signal of mental health (particularly emotion regulation, anxiety, and internalizing and externalizing disorders).[28][29]
The polyvagal theory by Porges is an influential model of how the vagal pathways respond to novelty and to stressful external stimuli.[30][31][32] The theory proposes that there are two vagal systems, one that is shared with reptiles and amphibia and a second, more recent, system that is unique to mammals. The two pathways behave differently and can work against each other. This theory can account for several psychophysiological phenomena and psychosomatic illnesses.[30][26] However, recent studies indicate that the vagal "system" described by Porges as being unique to mammals existed long before the evolution of mammals.[33][34]
RSA and vagal tone are used as markers to help determine the severity of neurodevelopmental disorders such as Autism Spectrum Disorders.[35] Children with more secure attachments with their mothers exhibited greater empathetic responsiveness, less social inhibition, and higher vagal tone.[36]
Other estimates of vagal tone
There are several methods of estimating vagal tone other than measuring RSA, including:
- Indexes of beat-to-beat variability such as RMSSD reported by The Task Force of the European Society of Cardiology and Heart Rhythm Society.[37] Frequency analysis of heart rate in the range 0.15–0.4 Hz has been reported to quantify vagal tone.[25][38]
- Computation of the "power spectrum", or the ratio between the low frequency and high frequency spectral components, of heart rate variability.[7][37] This has been used to measure the change in sympatho-vagal balance during hypnosis.[39]
See also
References
- Berntson GG, Cacioppo JT, Quigley KS (March 1993). "Respiratory sinus arrhythmia: autonomic origins, physiological mechanisms, and psychophysiological implications". Psychophysiology. 30 (2): 183–96. doi:10.1111/j.1469-8986.1993.tb01731.x. PMID 8434081.
- Diamond LM, Fagundes CP, Butterworth MR (2011). "Attachment Style, Vagal Tone, and Empathy During Mother-Adolescent Interactions". Journal of Research on Adolescence. 22 (1): 165–184. doi:10.1111/j.1532-7795.2011.00762.x.
- Grossman P, Wilhelm FH, Spoerle M (August 2004). "Respiratory sinus arrhythmia, cardiac vagal control, and daily activity". American Journal of Physiology. Heart and Circulatory Physiology. 287 (2): H728–34. doi:10.1152/ajpheart.00825.2003. PMID 14751862.
- Howland RH (June 2014). "Vagus Nerve Stimulation". Current Behavioral Neuroscience Reports. 1 (2): 64–73. doi:10.1007/s40473-014-0010-5. PMC 4017164. PMID 24834378.
- Porges SW, Doussard-Roosevelt JA, Maiti AK (2008). "Vagal tone and the physiological regulation of emotion". Monographs of the Society for Research in Child Development. 59 (2–3): 167–86. doi:10.1111/j.1540-5834.1994.tb01283.x. PMID 7984159.
- Brock C, Jessen N, Brock B, Jakobsen PE, Hansen TK, Rantanen JM, Riahi S, Dimitrova YK, Dons-Jensen A, Aziz Q, Drewes AM, Farmer AD (October 2017). "Cardiac vagal tone, a non-invasive measure of parasympathetic tone, is a clinically relevant tool in Type 1 diabetes mellitus". Diabetic Medicine. 34 (10): 1428–1434. doi:10.1111/dme.13421. PMID 28703868. S2CID 25030686.
- Montano N, Ruscone TG, Porta A, Lombardi F, Pagani M, Malliani A (October 1994). "Power spectrum analysis of heart rate variability to assess the changes in sympathovagal balance during graded orthostatic tilt". Circulation. 90 (4): 1826–31. doi:10.1161/01.CIR.90.4.1826. PMID 7923668.
- Nunan D, Sandercock GR, Brodie DA (November 2010). "A quantitative systematic review of normal values for short-term heart rate variability in healthy adults". Pacing and Clinical Electrophysiology. 33 (11): 1407–17. doi:10.1111/j.1540-8159.2010.02841.x. PMID 20663071. S2CID 44378765.
- Ludwig, Carl (1847). "On the influence of respiratory movements on blood flow in the aortic system [in German]". Arch Anat Physiol Leipzig. 13: 242–302.
- De Burgh Daly M (1985), "Interactions Between Respiration and Circulation", Comprehensive Physiology, John Wiley & Sons, Inc., pp. 529–594, doi:10.1002/cphy.cp030216, ISBN 9780470650714
- Hathorn MK (April 1987). "Respiratory sinus arrhythmia in new-born infants". The Journal of Physiology. 385: 1–12. doi:10.1113/jphysiol.1987.sp016480. PMC 1192333. PMID 3656159.
- Myers MM, Fifer W, Haiken J, Stark RI (June 1990). "Relationships between breathing activity and heart rate in fetal baboons". The American Journal of Physiology. 258 (6 Pt 2): R1479–85. doi:10.1152/ajpregu.1990.258.6.R1479. PMID 2360694.
- Hayano J, Yasuma F, Okada A, Mukai S, Fujinami T (August 1996). "Respiratory sinus arrhythmia. A phenomenon improving pulmonary gas exchange and circulatory efficiency". Circulation. 94 (4): 842–7. doi:10.1161/01.CIR.94.4.842. PMID 8772709.
- Castellini MA, Rea LD, Sanders JL, Castellini JM, Zenteno-Savin T (November 1994). "Developmental changes in cardiorespiratory patterns of sleep-associated apnea in northern elephant seals". The American Journal of Physiology. 267 (5 Pt 2): R1294–301. doi:10.1152/ajpregu.1994.267.5.R1294. PMID 7977857.
- Bartsch RP, Schumann AY, Kantelhardt JW, Penzel T, Ivanov PC (June 2012). "Phase transitions in physiologic coupling". Proceedings of the National Academy of Sciences of the United States of America. 109 (26): 10181–6. Bibcode:2012PNAS..10910181B. doi:10.1073/pnas.1204568109. PMC 3387128. PMID 22691492.
- Hayano J, Sakakibara Y, Yamada M, Kamiya T, Fujinami T, Yokoyama K, Watanabe Y, Takata K (March 1990). "Diurnal variations in vagal and sympathetic cardiac control". The American Journal of Physiology. 258 (3 Pt 2): H642–6. doi:10.1152/ajpheart.1990.258.3.H642. PMID 2316678.
- Porges SW (1986), "Respiratory Sinus Arrhythmia: Physiological Basis, Quantitative Methods, and Clinical Implications", Cardiorespiratory and Cardiosomatic Psychophysiology, Springer US, pp. 101–115, doi:10.1007/978-1-4757-0360-3_7, ISBN 9781475703627
- Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, Sandrone G, Malfatto G, Dell'Orto S, Piccaluga E (August 1986). "Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog". Circulation Research. 59 (2): 178–93. doi:10.1161/01.RES.59.2.178. PMID 2874900.
- Grossman P, van Beek J, Wientjes C (November 1990). "A comparison of three quantification methods for estimation of respiratory sinus arrhythmia". Psychophysiology. 27 (6): 702–14. doi:10.1111/j.1469-8986.1990.tb03198.x. PMID 2100356.
- Dick TE, Hsieh YH, Dhingra RR, Baekey DM, Galán RF, Wehrwein E, Morris KF (2014). Cardiorespiratory coupling: common rhythms in cardiac, sympathetic, and respiratory activities. Progress in Brain Research. 209. Elsevier. pp. 191–205. doi:10.1016/b978-0-444-63274-6.00010-2. ISBN 9780444632746. PMC 4052709. PMID 24746049.
- Grossman, Paul; Taylor, Edwin W. (2007). "Toward understanding respiratory sinus arrhythmia: relations to cardiac vagal tone, evolution and biobehavioral functions". Biological Psychology. 74 (2): 263–285. doi:10.1016/j.biopsycho.2005.11.014. ISSN 0301-0511. PMID 17081672. S2CID 16818862.
- Hayano, Junichiro; Yasuma, Fumihiko (2003-04-01). "Hypothesis: respiratory sinus arrhythmia is an intrinsic resting function of cardiopulmonary system". Cardiovascular Research. 58 (1): 1–9. doi:10.1016/s0008-6363(02)00851-9. ISSN 0008-6363. PMID 12667941.
- Ben-Tal A, Shamailov SS, Paton JF (April 2012). "Evaluating the physiological significance of respiratory sinus arrhythmia: looking beyond ventilation-perfusion efficiency". The Journal of Physiology. 590 (8): 1989–2008. doi:10.1113/jphysiol.2011.222422. PMC 3573317. PMID 22289913.
- Hayano J, Yasuma F, Okada A, Mukai S, Fujinami T (August 1996). "Respiratory sinus arrhythmia. A phenomenon improving pulmonary gas exchange and circulatory efficiency". Circulation. 94 (4): 842–7. doi:10.1161/01.cir.94.4.842. PMID 8772709.
- Lewis GF, Furman SA, McCool MF, Porges SW (February 2012). "Statistical strategies to quantify respiratory sinus arrhythmia: are commonly used metrics equivalent?". Biological Psychology. 89 (2): 349–64. doi:10.1016/j.biopsycho.2011.11.009. PMC 3269511. PMID 22138367.
- Graziano P, Derefinko K (September 2013). "Cardiac vagal control and children's adaptive functioning: a meta-analysis". Biological Psychology. 94 (1): 22–37. doi:10.1016/j.biopsycho.2013.04.011. PMC 4074920. PMID 23648264.
- Masi CM, Hawkley LC, Rickett EM, Cacioppo JT (February 2007). "Respiratory sinus arrhythmia and diseases of aging: obesity, diabetes mellitus, and hypertension". Biological Psychology. 74 (2): 212–23. doi:10.1016/j.biopsycho.2006.07.006. PMC 1804292. PMID 17034928.
- Connell AM, Hughes-Scalise A, Klostermann S, Azem T (October 2011). "Maternal depression and the heart of parenting: respiratory sinus arrhythmia and affective dynamics during parent-adolescent interactions". Journal of Family Psychology. 25 (5): 653–62. doi:10.1037/a0025225. PMID 21875198.
- Porges, SW; Doussard-Roosevelt, JA; Maiti, AK (1994). "Vagal tone and the physiological regulation of emotion". Monographs of the Society for Research in Child Development. 59 (2–3): 167–86. doi:10.1111/j.1540-5834.1994.tb01283.x. ISSN 0037-976X. JSTOR 1166144. PMID 7984159.
A review of research indicates that baseline levels of cardiac vagal tone and vagal tone reactivity abilities are associated with behavioral measures of reactivity, the expression of emotion, and self-regulation skills. Thus, we propose that cardiac vagal tone can serve as an index of emotion regulation. Historically, the vagus and other components of the parasympathetic nervous system have not been incorporated in theories of emotion.
- Porges SW (July 1995). "Orienting in a defensive world: mammalian modifications of our evolutionary heritage. A Polyvagal Theory". Psychophysiology. 32 (4): 301–18. doi:10.1111/j.1469-8986.1995.tb01213.x. PMID 7652107.
- Porges SW (August 2003). "The Polyvagal Theory: phylogenetic contributions to social behavior". Physiology & Behavior. 79 (3): 503–13. doi:10.1016/S0031-9384(03)00156-2. PMID 12954445. S2CID 14074575.
- Porges SW (December 2003). "Social engagement and attachment: a phylogenetic perspective". Annals of the New York Academy of Sciences. 1008 (1): 31–47. Bibcode:2003NYASA1008...31P. doi:10.1196/annals.1301.004. PMID 14998870. S2CID 1377353.
- Monteiro, Diana (2018). "Cardiorespiratory interactions previously identified as mammalian are present in the primitive lungfish". Science Advances. 4 (2): eaaq0800. Bibcode:2018SciA....4..800M. doi:10.1126/sciadv.aaq0800. PMC 5833999. PMID 29507882.
- Taylor, E. W. (2010). "Autonomic control of cardiorespiratory interactions in fish, amphibians and reptiles". Brazilian Journal of Medical and Biological Research. 43 (7): 600–610. doi:10.1590/S0100-879X2010007500044. PMID 20464342.
- Patriquin MA, Scarpa A, Friedman BH, Porges SW (March 2013). "Respiratory sinus arrhythmia: a marker for positive social functioning and receptive language skills in children with autism spectrum disorders". Developmental Psychobiology. 55 (2): 101–12. doi:10.1002/dev.21002. PMID 22212893.
- Diamond LM, Fagundes CP, Butterworth MR (2012). "Attachment style, vagal tone, and empathy during mother–adolescent interactions". Journal of Research on Adolescence. 22 (1): 165–184. doi:10.1111/j.1532-7795.2011.00762.x.
- Malik, M.; Bigger, J. T.; Camm, A. J.; Kleiger, R. E.; Malliani, A.; Moss, A. J.; Schwartz, P. J. (March 1996). "Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology". European Heart Journal. 17 (3): 354–81. doi:10.1093/oxfordjournals.eurheartj.a014868. PMID 8737210.
- Myers MM, Elliott AJ, Odendaal HJ, Burd L, Angal J, Groenewald C, Nugent JD, Yang JS, Isler JR, Dukes KA, Robinson F, Fifer WP (August 2017). "Cardiorespiratory physiology in the safe passage study: protocol, methods and normative values in unexposed infants". Acta Paediatrica. 106 (8): 1260–1272. doi:10.1111/apa.13873. PMC 5530586. PMID 28419567.
- DeBenedittis G, Cigada M, Bianchi A, Signorini MG, Cerutti S (April 1994). "Autonomic changes during hypnosis: a heart rate variability power spectrum analysis as a marker of sympatho-vagal balance". The International Journal of Clinical and Experimental Hypnosis. 42 (2): 140–52. doi:10.1080/00207149408409347. PMID 8200716.