Voacamine
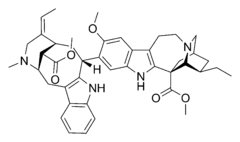
Voacamine, also known under the older names voacanginine and vocamine, is a naturally occurring dimeric indole alkaloid of the secologanin type, found in a number of plants, including Voacanga africana and Tabernaemontana divaricata.
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| ECHA InfoCard | 100.020.139 |
| Chemical and physical data | |
| Formula | C43H52N4O5 |
| Molar mass | 704.912 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
It is approved for use as an antimalarial drug in several African countries.[1]
Voacamine exhibits cannabinoid CB1 receptor antagonistic activity.[2]
Chemical structure
There is considerable confusion about the absolute stereochemical configuration of voacamine and the originally published absolute structure had to be later revised.[3][4]
Toxicity/Adverse Effect/Side Effect
Voacamine can cause hypertension in high dose.[5]
References
- "Voacamine". DrugBank. Canadian Institutes of Health Research.
- Kitajima M, Iwai M, Kikura-Hanajiri R, Goda Y, Iida M, Yabushita H, Takayama H (April 2011). "Discovery of indole alkaloids with cannabinoid CB1 receptor antagonistic activity". Bioorganic & Medicinal Chemistry Letters. 21 (7): 1962–4. doi:10.1016/j.bmcl.2011.02.036. PMID 21376588.
- Kutney JP, Brown RT, Piers E (March 1966). "The absolute configuration of the Iboga alkaloids". Canadian Journal of Chemistry. 44 (5): 637–9. doi:10.1139/v66-087.
- Kutney JP, Fuji K, Treasurywala AM, Fayos J, Clardy J, Scott AI, Wei CC (1973). "Structure and Absolute Configuration of (+)-Coronaridine Hydrobromide. A Comment on the Absolute Configuration of the Iboga Alkaloids". J. Am. Chem. Soc. 95 (16): 5407–5409. doi:10.1021/ja00797a049.
- Leeuwenberg AJ (1985). "oacanga, (Apocynaceae), a review of its taxonomy, phytochemistry, ethnobotany and pharmacology". Agric. Univ. Wagenigen. pp. 85–3.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.