β-Lactamase inhibitor
Beta-lactamases are a family of enzymes involved in bacterial resistance to beta-lactam antibiotics. They act by breaking the beta-lactam ring that allows penicillin-like antibiotics to work. Strategies for combating this form of resistance have included the development of new beta-lactam antibiotics that are more resistant to cleavage and the development of the class of enzyme inhibitors called beta-lactamase inhibitors.[1] Although β-lactamase inhibitors have little antibiotic activity of their own,[2] they prevent bacterial degradation of beta-lactam antibiotics and thus extend the range of bacteria the drugs are effective against.
Medical uses
The most important use of beta-lactamase inhibitors is in the treatment of infections known or believed to be caused by gram-negative bacteria, as beta-lactamase production is an important contributor to beta-lactam resistance in these pathogens. In contrast, most beta-lactam resistance in gram-positive bacteria is due to variations in penicillin-binding proteins that lead to reduced binding to the beta-lactam.[3][4] The gram-positive pathogen Staphylococcus aureus produces beta-lactamases, but beta-lactamase inhibitors play a lesser role in treatment of these infections because the most resistant strains (methicillin-resistant Staphylococcus aureus) also use variant penicillin-binding proteins.[5][6]
Mechanism of action
The Ambler classification system groups known beta-lactamase enzymes into four groups according to sequence homology and presumed phylogenetic relationships. Classes A, C and D cleave beta-lactams by a multi-step mechanism analogous to the mechanism of serine proteases. Upon binding, a serine hydroxyl group in the beta-lactamase active site forms a transient covalent bond to the beta-lactam ring carbonyl group, cleaving the beta-lactam ring in the process. In a second step, nucleophilic attack by a water molecule cleaves the covalent bond between the enzyme and the carbonyl group of the erstwhile beta-lactam. This allows the degraded beta-lactam to diffuse away and frees up the enzyme to process additional beta-lactam molecules.
Currently available beta-lactamase inhibitors are effective against Ambler Class A beta-lactamases (tazobactam, clavulanate, and sulbactam) or against Ambler Class A, C and some Class D beta-lactamases (avibactam). Like beta-lactam antibiotics, they are processed by beta-lactamases to form an initial covalent intermediate. Unlike the case of beta-lactam antibiotics, the inhibitors act as suicide substrates (tazobactam and sulbactam) which ultimately leads to the degradation of the beta-lactamase.[7] Avibactam on the other hand does not contain a beta-lactam ring (non beta-lactam beta-lactamase inhibitor), and instead binds reversibly.[8][9]
Ambler Class B beta-lactamases cleave beta-lactams by a mechanism similar to that of metalloproteases. As no covalent intermediate is formed, the mechanism of action of marketed beta-lactamase inhibitors is not applicable. Thus the spread of bacterial strains expressing metallo beta-lactamases such as the New Delhi metallo-beta-lactamase 1 has engendered considerable concern.[10]
Commonly used agents
Currently marketed β-lactamase inhibitors are not sold as individual drugs. Instead they are co-formulated with a β-lactam antibiotic with a similar serum half-life. This is done not only for dosing convenience, but also to minimize resistance development that might occur as a result of varying exposure to one or the other drug. The main classes of β-lactam antibiotics used to treat gram-negative bacterial infections include (in approximate order of intrinsic resistance to cleavage by β-lactamases) penicillins (especially aminopenicillins and ureidopenicillins), 3rd generation cephalosporins, and carbapenems. Individual β-lactamase variants may target one or many of these drug classes, and only a subset will be inhibited by a given β-lactamase inhibitor.[9] β-lactamase inhibitors expand the useful spectrum of these β-lactam antibiotics by inhibiting the β-lactamase enzymes produced by bacteria to deactivate them.[11]
- β-lactamase inhibitors with a β-lactam core:
- Tebipenem is the first carbapenem to be administered orally in the form of tebipenem-pivoxil. Structural and kinetic studies of tebipenem is available with M. tuberculosis beta-lactamase (BlaC).[12]
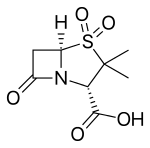
- Clavulanic acid or clavulanate, usually combined with amoxicillin (Augmentin) or ticarcillin (Timentin)
- Sulbactam, usually combined with ampicillin (Unasyn) or cefoperazone (Sulperazon)
- Tazobactam, usually combined with piperacillin (Zosyn and Tazocin)
- β-lactamase inhibitors without a β-lactam core:
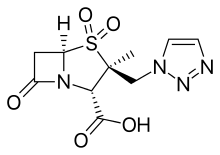
- Avibactam, approved in combination with ceftazidime (Avycaz), currently undergoing clinical trials for combination with ceftaroline
- Relebactam, used in combination with imipenem/cilastatin (Recarbrio).[13][14]
- Vaborbactam, used in combination with meropenem (Vabomere)[15]
Beta-lactamase producing bacteria
Bacteria that can produce beta-lactamases include, but are not limited to:
- Staphylococcus
- MRSA(Methicillin-resistant Staphylococcus aureus)
- Enterobacteriaceae:
- Haemophilus influenzae
- Neisseria gonorrhoeae
- Pseudomonas aeruginosa
- Mycobacterium tuberculosis
Research
Some bacteria can produce extended spectrum β-lactamases (ESBLs) making the infection more difficult to treat and conferring additional resistance to penicillins, cephalosporins, and monobactams.[16] Boronic acid derivatives are currently under vast and extensive research as novel active site inhibitors for beta-lactamases because they contain a site that mimics the transition state that beta-lactams go through when undergoing hydrolysis via beta-lactamases. They have been found generally to fit well into the active site of many beta-lactamases and have the convenient property of being unable to be hydrolysed, and therefore rendered useless. This is a favorable drug design over many clinically used competing agents, because most of them, such as clavulanic acid, become hydrolysed, and are therefore only useful for a finite period of time. This generally causes the need for a higher concentration of competitive inhibitor than would be necessary in an unhydrolyzable inhibitor. Different boronic acid derivatives have the potential to be tailored to the many different isoforms of beta-lactamases, and therefore have the potential to reestablish potency of beta-lactam antibiotics.[17]
References
- Essack SY (October 2001). "The development of beta-lactam antibiotics in response to the evolution of beta-lactamases". Pharmaceutical Research. 18 (10): 1391–9. doi:10.1023/a:1012272403776. PMID 11697463. S2CID 34318096.
- "Beta-Lactamase Inhibitors". Department of Nursing of the Fort Hays State University College of Health and Life Sciences. October 2000. Archived from the original on 2007-09-27. Retrieved 2007-08-17.
- Georgopapadakou NH (October 1993). "Penicillin-binding proteins and bacterial resistance to beta-lactams". Antimicrobial Agents and Chemotherapy. 37 (10): 2045–53. doi:10.1128/aac.37.10.2045. PMC 192226. PMID 8257121.
- Zapun A, Contreras-Martel C, Vernet T (March 2008). "Penicillin-binding proteins and beta-lactam resistance". FEMS Microbiology Reviews. 32 (2): 361–85. doi:10.1111/j.1574-6976.2007.00095.x. PMID 18248419.
- Curello J, MacDougall C (July 2014). "Beyond Susceptible and Resistant, Part II: Treatment of Infections Due to Gram-Negative Organisms Producing Extended-Spectrum β-Lactamases". The Journal of Pediatric Pharmacology and Therapeutics. 19 (3): 156–64. doi:10.5863/1551-6776-19.3.156. PMC 4187532. PMID 25309145.
- Wolter DJ, Lister PD (2013). "Mechanisms of β-lactam resistance among Pseudomonas aeruginosa". Current Pharmaceutical Design. 19 (2): 209–22. doi:10.2174/13816128130203. PMID 22894618.
- Patrick, Graham L. (2017). An introduction to medicinal chemistry (6th ed.). Oxford, United Kingdom. ISBN 9780198749691. OCLC 987051883.
- Lahiri SD, Mangani S, Durand-Reville T, Benvenuti M, De Luca F, Sanyal G, Docquier JD (June 2013). "Structural insight into potent broad-spectrum inhibition with reversible recyclization mechanism: avibactam in complex with CTX-M-15 and Pseudomonas aeruginosa AmpC β-lactamases". Antimicrobial Agents and Chemotherapy. 57 (6): 2496–505. doi:10.1128/AAC.02247-12. PMC 3716117. PMID 23439634.
- Drawz SM, Bonomo RA (January 2010). "Three decades of beta-lactamase inhibitors". Clinical Microbiology Reviews. 23 (1): 160–201. doi:10.1128/CMR.00037-09. PMC 2806661. PMID 20065329.
- Biedenbach D, Bouchillon S, Hackel M, Hoban D, Kazmierczak K, Hawser S, Badal R (February 2015). "Dissemination of NDM metallo-β-lactamase genes among clinical isolates of Enterobacteriaceae collected during the SMART global surveillance study from 2008 to 2012". Antimicrobial Agents and Chemotherapy. 59 (2): 826–30. doi:10.1128/AAC.03938-14. PMC 4335866. PMID 25403666.
- Watson ID, Stewart MJ, Platt DJ (September 1988). "Clinical pharmacokinetics of enzyme inhibitors in antimicrobial chemotherapy". Clinical Pharmacokinetics. 15 (3): 133–64. doi:10.2165/00003088-198815030-00001. PMID 3052984. S2CID 2388750.
- Hazra S, Xu H, Blanchard JS (June 2014). "Tebipenem, a new carbapenem antibiotic, is a slow substrate that inhibits the β-lactamase from Mycobacterium tuberculosis". Biochemistry. 53 (22): 3671–8. doi:10.1021/bi500339j. PMC 4053071. PMID 24846409.
- "FDA approves new treatment for complicated urinary tract and complicated intra-abdominal infections". Food and Drug Administration. July 17, 2019.
- "Cilastatin/imipenem/relebactam — AdisInsight". Springer International Publishing AG. Retrieved 29 April 2016.
- "FDA approves new antibacterial drug" (Press release). Food and Drug Administration. August 29, 2017.
- Livermore DM (October 1995). "beta-Lactamases in laboratory and clinical resistance". Clinical Microbiology Reviews. 8 (4): 557–84. doi:10.1128/cmr.8.4.557. PMC 172876. PMID 8665470.
- Leonard DA, Bonomo RA, Powers RA (November 2013). "Class D β-lactamases: a reappraisal after five decades". Accounts of Chemical Research. 46 (11): 2407–15. doi:10.1021/ar300327a. PMC 4018812. PMID 23902256.
External links
- Xu H, Hazra S, Blanchard JS (June 2012). "NXL104 irreversibly inhibits the β-lactamase from Mycobacterium tuberculosis". Biochemistry. 51 (22): 4551–7. doi:10.1021/bi300508r. PMC 3448018. PMID 22587688.
- Kurz SG, Wolff KA, Hazra S, Bethel CR, Hujer AM, Smith KM, Xu Y, Tremblay LW, Blanchard JS, Nguyen L, Bonomo RA (December 2013). "Can inhibitor-resistant substitutions in the Mycobacterium tuberculosis β-Lactamase BlaC lead to clavulanate resistance?: a biochemical rationale for the use of β-lactam-β-lactamase inhibitor combinations". Antimicrobial Agents and Chemotherapy. 57 (12): 6085–96. doi:10.1128/AAC.01253-13. PMC 3837893. PMID 24060876.