6-phosphogluconolactonase
6-Phosphogluconolactonase (6PGL, PGLS) is a cytosolic enzyme found in all organisms that catalyzes the hydrolysis of 6-phosphogluconolactone to 6-phosphogluconic acid in the oxidative phase of the pentose phosphate pathway.[2] The tertiary structure of 6PGL employs an α/β hydrolase fold, with active site residues clustered on the loops of the α-helices. Based on the crystal structure of the enzyme, the mechanism is proposed to be dependent on proton transfer by a histidine residue in the active site.[1] 6PGL selectively catalyzes the hydrolysis of δ-6-phosphogluconolactone, and has no activity on the γ isomer.[3]
| 6-phosphogluconolactonase | |||||||
|---|---|---|---|---|---|---|---|
 Crystallized monomer of 6-phosphogluconolactonase from Trypanosoma brucei complexed with 6-phosphogluconic acid[1] | |||||||
| Identifiers | |||||||
| Symbol | PGLS | ||||||
| NCBI gene | 25796 | ||||||
| HGNC | 8903 | ||||||
| OMIM | 604951 | ||||||
| RefSeq | NM_012088 | ||||||
| UniProt | O95336 | ||||||
| Other data | |||||||
| EC number | 3.1.1.31 | ||||||
| Locus | Chr. 19 p13.2 | ||||||
| |||||||
Enzyme Mechanism
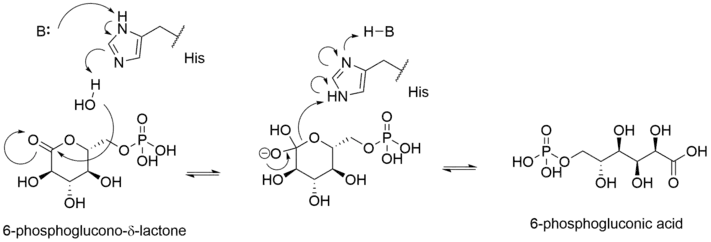
6PGL hydrolysis of 6-phosphogluconolactone to 6-phosphogluconic acid has been proposed to proceed via proton transfer to the O5 ring oxygen atom,[4] similar to xylose isomerase[5] and ribose-5-phosphate isomerase.[6] The reaction initiates via attack of a hydroxide ion at the C5 ester. A tetrahedral intermediate forms and elimination of the ester linkage follows, aided by donation of a proton from an active site histidine residue. The specific residue that participates in the proton transfer eluded researchers until 2009, as previous structural studies demonstrated two possible conformations of the substrate in the active site, which position the O5 ring oxygen proximal to either an arginine or a histidine residue.[1] Molecular dynamic simulations were employed to discover that the residue that donates a proton is histidine, and that the arginine residues are only involved in electric stabilization of the negatively charged phosphate group.[4] Electric stabilization of the enzyme-substrate complex also occurs between the product carboxylate and backbone amines of surrounding glycine residues.[4]
Enzyme Structure
6PGL in Homo sapiens exists as a monomer at cytosolic physiological conditions, and is composed of 258 amino acid residues with a total molecular mass of ~30 kDa.[7] The tertiary structure of the enzyme utilizes an α/β hydrolase fold, with both parallel and anti-parallel β-sheets surrounded by eight α-helices and five 310 helices.[1] Stability of the tertiary structure of the protein is reinforced through salt bridges between aspartic acid and arginine residues, and from aromatic side-chain stacking interactions.[1] 6PGL isolated from Trypanosoma brucei was found to bind with a Zn+2 ion in a non-catalytic role, but this has not been observed in other organisms, including Thermotoga maritima and Vibrio cholerae.[1]
Biological Function
6-phosphogluconolactonase catalyzes the conversion of 6-phosphogluconolactone to 6-phosphogluconic acid, both intermediates in the oxidative phase of the pentose phosphate pathway, in which glucose is converted into ribulose 5-phosphate. The oxidative phase of the pentose phosphate pathway releases CO2 and results in the generation of two equivalents of NADPH from NADP+. The final product, ribulose 5-phosphate, is further processed by the organism during the non-oxidative phase of the pentose phosphate pathway to synthesize biomolecules including nucleotides, ATP, and Coenzyme A.[2]
The enzyme that precedes 6PGL in the pentose phosphate pathway, glucose-6-phosphate dehydrogenase, exclusively forms the δ-isomer of 6-phosphogluconolactone. However, if accumulated, this compound can undergo intramolecular rearrangement to isomerize to the more stable γ-form, which is unable to be hydrolyzed by 6PGL and cannot continue to the non-oxidative phase of the pentose phosphate pathway. By quickly hydrolyzing the δ-isomer of 6-phosphogluconolactone, 6PGL prevents its accumulation and subsequent formation of the γ-isomer, which would be wasteful of the glucose resources available to the cell.[3] 6-phosphogluconolactone is also susceptible to attack from intracellular nucleophiles, evidenced by α-N-6-phosphogluconoylation of His-tagged proteins expressed in E. coli,[8][9] and efficient hydrolysis of 6-phosphogluconolactone by 6PGL prevents lactone accumulation and consequent toxic reactions from occurring between the lactone intermediate and the cell.[3]
Disease Relevance
Malarial parasites Plasmodium berghei and Plasmodium falciparum have been shown to express a bi-functional enzyme that exhibits both glucose-6-phosphate dehydrogenase and 6-phosphogluconolactonase activity, enabling it to catalyze the first two steps of the pentose phosphate pathway.[10] This bifunctional enzyme has been identified as a druggable target for malarial parasites,[11] and high-throughput screening of small molecule inhibitors has resulted in the discovery of novel compounds that can potentially be translated into potent antimalarials.[12][13]
References
- Delarue M, Duclert-Savatier N, Miclet E, Haouz A, Giganti D, Ouazzani J, Lopez P, Nilges M, Stoven V (February 2007). "Three dimensional structure and implications for the catalytic mechanism of 6-phosphogluconolactonase from Trypanosoma brucei". Journal of Molecular Biology. 366 (3): 868–81. doi:10.1016/j.jmb.2006.11.063. PMID 17196981.
- Berg J, Tymoczko J, Stryer L (2012). Biochemistry (Seventh ed.). New York, NY 10010: W.H. Freeman and Company. pp. 600–601. ISBN 9781429229364.CS1 maint: location (link)
- Miclet E, Stoven V, Michels PA, Opperdoes FR, Lallemand JY, Duffieux F (September 2001). "NMR spectroscopic analysis of the first two steps of the pentose-phosphate pathway elucidates the role of 6-phosphogluconolactonase". The Journal of Biological Chemistry. 276 (37): 34840–6. doi:10.1074/jbc.M105174200. PMID 11457850.
- Duclert-Savatier N, Poggi L, Miclet E, Lopes P, Ouazzani J, Chevalier N, Nilges M, Delarue M, Stoven V (May 2009). "Insights into the enzymatic mechanism of 6-phosphogluconolactonase from Trypanosoma brucei using structural data and molecular dynamics simulation". Journal of Molecular Biology. 388 (5): 1009–21. doi:10.1016/j.jmb.2009.03.063. PMID 19345229.
- Whitlow M, Howard AJ, Finzel BC, Poulos TL, Winborne E, Gilliland GL (1991-03-01). "A metal-mediated hydride shift mechanism for xylose isomerase based on the 1.6 A Streptomyces rubiginosus structures with xylitol and D-xylose". Proteins. 9 (3): 153–73. doi:10.1002/prot.340090302. PMID 2006134.
- Zhang RG, Andersson CE, Savchenko A, Skarina T, Evdokimova E, Beasley S, Arrowsmith CH, Edwards AM, Joachimiak A, Mowbray SL (January 2003). "Structure of Escherichia coli ribose-5-phosphate isomerase: a ubiquitous enzyme of the pentose phosphate pathway and the Calvin cycle". Structure. 11 (1): 31–42. doi:10.1016/S0969-2126(02)00933-4. PMC 2792023. PMID 12517338.
- Collard F, Collet JF, Gerin I, Veiga-da-Cunha M, Van Schaftingen E (October 1999). "Identification of the cDNA encoding human 6-phosphogluconolactonase, the enzyme catalyzing the second step of the pentose phosphate pathway(1)". FEBS Letters. 459 (2): 223–6. doi:10.1016/S0014-5793(99)01247-8. PMID 10518023. S2CID 29302175.
- Geoghegan KF, Dixon HB, Rosner PJ, Hoth LR, Lanzetti AJ, Borzilleri KA, Marr ES, Pezzullo LH, Martin LB, LeMotte PK, McColl AS, Kamath AV, Stroh JG (February 1999). "Spontaneous alpha-N-6-phosphogluconoylation of a "His tag" in Escherichia coli: the cause of extra mass of 258 or 178 Da in fusion proteins". Analytical Biochemistry. 267 (1): 169–84. doi:10.1006/abio.1998.2990. PMID 9918669.
- Kim KM, Yi EC, Baker D, Zhang KY (May 2001). "Post-translational modification of the N-terminal His tag interferes with the crystallization of the wild-type and mutant SH3 domains from chicken src tyrosine kinase". Acta Crystallographica Section D. 57 (Pt 5): 759–62. doi:10.1107/s0907444901002918. PMID 11320329.
- Clarke JL, Scopes DA, Sodeinde O, Mason PJ (April 2001). "Glucose-6-phosphate dehydrogenase-6-phosphogluconolactonase. A novel bifunctional enzyme in malaria parasites". European Journal of Biochemistry. 268 (7): 2013–9. doi:10.1046/j.1432-1327.2001.02078.x. PMID 11277923.
- Allen SM, Lim EE, Jortzik E, Preuss J, Chua HH, MacRae JI, Rahlfs S, Haeussler K, Downton MT, McConville MJ, Becker K, Ralph SA (October 2015). "Plasmodium falciparum glucose-6-phosphate dehydrogenase 6-phosphogluconolactonase is a potential drug target". The FEBS Journal. 282 (19): 3808–23. doi:10.1111/febs.13380. PMID 26198663. S2CID 46398163.
- Preuss J, Hedrick M, Sergienko E, Pinkerton A, Mangravita-Novo A, Smith L, Marx C, Fischer E, Jortzik E, Rahlfs S, Becker K, Bode L (July 2012). "High-throughput screening for small-molecule inhibitors of plasmodium falciparum glucose-6-phosphate dehydrogenase 6-phosphogluconolactonase". Journal of Biomolecular Screening. 17 (6): 738–51. doi:10.1177/1087057112442382. PMID 22496096.
- Preuss J, Maloney P, Peddibhotla S, Hedrick MP, Hershberger P, Gosalia P, Milewski M, Li YL, Sugarman E, Hood B, Suyama E, Nguyen K, Vasile S, Sergienko E, Mangravita-Novo A, Vicchiarelli M, McAnally D, Smith LH, Roth GP, Diwan J, Chung TD, Jortzik E, Rahlfs S, Becker K, Pinkerton AB, Bode L (August 2012). "Discovery of a Plasmodium falciparum glucose-6-phosphate dehydrogenase 6-phosphogluconolactonase inhibitor (R,Z)-N-((1-ethylpyrrolidin-2-yl)methyl)-2-(2-fluorobenzylidene)-3-oxo-3,4-dihydro-2H-benzo[b][1,4]thiazine-6-carboxamide (ML276) that reduces parasite growth in vitro". Journal of Medicinal Chemistry. 55 (16): 7262–72. doi:10.1021/jm300833h. PMC 3530835. PMID 22813531.
External links
- 6-phosphogluconolactonase at the US National Library of Medicine Medical Subject Headings (MeSH)
- Collard F, Collet JF, Gerin I, Veiga-da-Cunha M, Van Schaftingen E (October 1999). "Identification of the cDNA encoding human 6-phosphogluconolactonase, the enzyme catalyzing the second step of the pentose phosphate pathway(1)". FEBS Letters. 459 (2): 223–6. doi:10.1016/S0014-5793(99)01247-8. PMID 10518023. S2CID 29302175.