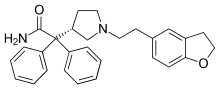
Darifenacin
Darifenacin (trade name Enablex in United States and Canada, Emselex in the European Union) is a medication used to treat urinary incontinence. It was discovered by scientists at the Pfizer research site in Sandwich, UK under the identifier UK-88,525 and used to be marketed by Novartis. In 2010, the US rights were sold to Warner Chilcott for US$400 million .
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Enablex, Emselex |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605039 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 15 to 19% (dose-dependent) |
| Protein binding | 98% |
| Metabolism | Liver (CYP2D6- and CYP3A4-mediated) |
| Elimination half-life | 13 to 19 hours |
| Excretion | Kidney (60%) and biliary (40%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.118.382 |
| Chemical and physical data | |
| Formula | C28H30N2O2 |
| Molar mass | 426.560 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Adverse effects
Darifenacin should not be used in people with urinary retention. Anticholinergic agents, such as darifenacin, may also produce constipation and blurred vision. Heat prostration (due to decreased sweating) can occur when anticholinergics such as darifenacin are used in a hot environment.[1]
Medical uses
Darifenacin is indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency in adults. It may also be recommended with an alpha blocker to help provide symptomatic benefit for overactive bladder and obstructive symptoms such as likely associated with benign prostatic hypertrophy. [2]
Mechanism of action
Darifenacin works by blocking the M3 muscarinic acetylcholine receptor, which is primarily responsible for bladder muscle contractions. It thereby decreases the urgency to urinate. It is not known whether this selectivity for the M3 receptor translates into any clinical advantage when treating symptoms of overactive bladder syndrome.[1]
References
- "Enablex- darifenacin tablet, extended release". DailyMed. 24 September 2016. Retrieved 22 October 2020.
- American Urological Association (AUA) Guideline. Diagnosis and Treatment of Overactive Bladder in Adults: AUA/SUFA guideline 2012
External links
- "Darifenacin". Drug Information Portal. U.S. National Library of Medicine.
- "Darifenacin hydrobromide". Drug Information Portal. U.S. National Library of Medicine.