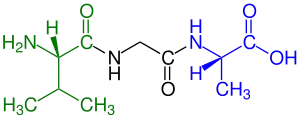Peptide
Peptides (from Greek language πεπτός, peptós "digested"; derived from πέσσειν, péssein "to digest") are short chains of between two and fifty amino acids, linked by peptide bonds.[1][2] Chains of fewer than ten or fifteen amino acids are called oligopeptides, and include dipeptides, tripeptides, and tetrapeptides.

blue marked carboxyl end (L-Alanine).
A polypeptide is a longer, continuous, unbranched peptide chain of up to approximately fifty amino acids.[3] Hence, peptides fall under the broad chemical classes of biological polymers and oligomers, alongside nucleic acids, oligosaccharides, polysaccharides, and others.
A polypeptide that contains more than approximately fifty amino acids is known as a protein.[3][4][5] Proteins consist of one or more polypeptides arranged in a biologically functional way, often bound to ligands such as coenzymes and cofactors, or to another protein or other macromolecule such as DNA or RNA, or to complex macromolecular assemblies.[6]
Amino acids that have been incorporated into peptides are termed residues. A water molecule is released during formation of each amide bond.[7] All peptides except cyclic peptides have an N-terminal (amine group) and C-terminal (carboxyl group) residue at the end of the peptide (as shown for the tetrapeptide in the image).
Classes
Many kinds of peptides are known. They have been classified or categorized according to their sources and functions. According to the Handbook of Biologically Active Peptides, some groups of peptides include plant peptides, bacterial/antibiotic peptides, fungal peptides, invertebrate peptides, amphibian/skin peptides, venom peptides, cancer/anticancer peptides, vaccine peptides, immune/inflammatory peptides, brain peptides, endocrine peptides, ingestive peptides, gastrointestinal peptides, cardiovascular peptides, renal peptides, respiratory peptides, opiate peptides, neurotrophic peptides, and blood–brain peptides.[8]
Some ribosomal peptides are subject to proteolysis. These function, typically in higher organisms, as hormones and signaling molecules. Some organisms produce peptides as antibiotics, such as microcins and bacteriocins.[9]
Peptides frequently have post-translational modifications such as phosphorylation, hydroxylation, sulfonation, palmitoylation, glycosylation, and disulfide formation. In general, peptides are linear, although lariat structures have been observed.[10] More exotic manipulations do occur, such as racemization of L-amino acids to D-amino acids in platypus venom.[11]
Nonribosomal peptides are assembled by enzymes, not the ribosome. A common non-ribosomal peptide is glutathione, a component of the antioxidant defenses of most aerobic organisms.[12] Other nonribosomal peptides are most common in unicellular organisms, plants, and fungi and are synthesized by modular enzyme complexes called nonribosomal peptide synthetases.[13]
These complexes are often laid out in a similar fashion, and they can contain many different modules to perform a diverse set of chemical manipulations on the developing product.[14] These peptides are often cyclic and can have highly complex cyclic structures, although linear nonribosomal peptides are also common. Since the system is closely related to the machinery for building fatty acids and polyketides, hybrid compounds are often found. The presence of oxazoles or thiazoles often indicates that the compound was synthesized in this fashion.[15]
Peptones are derived from animal milk or meat digested by proteolysis.[16] In addition to containing small peptides, the resulting material includes fats, metals, salts, vitamins, and many other biological compounds. Peptones are used in nutrient media for growing bacteria and fungi.[17]
Peptide fragments refer to fragments of proteins that are used to identify or quantify the source protein.[18] Often these are the products of enzymatic degradation performed in the laboratory on a controlled sample, but can also be forensic or paleontological samples that have been degraded by natural effects.[19][20]
Chemical synthesis

Example families
The peptide families in this section are ribosomal peptides, usually with hormonal activity. All of these peptides are synthesized by cells as longer "propeptides" or "proproteins" and truncated prior to exiting the cell. They are released into the bloodstream where they perform their signaling functions.
Antimicrobial peptides
- Magainin family
- Cecropin family
- Cathelicidin family
- Defensin family
Tachykinin peptides
Vasoactive intestinal peptides
Pancreatic polypeptide-related peptides
Opioid peptides
- Proopiomelanocortin (POMC) peptides
- Enkephalin pentapeptides
- Prodynorphin peptides
Calcitonin peptides
Self-Assembling peptides
Other peptides
- B-type Natriuretic Peptide (BNP) - produced in the myocardium and useful in medical diagnosis
- Lactotripeptides - Lactotripeptides might reduce blood pressure,[29][30][31] although the evidence is mixed.[32]
- Peptidic components from traditional Chinese medicine Colla Corii Asini in hematopoiesis.[33]
Terminology
Length
Several terms related to peptides have no strict length definitions, and there is often overlap in their usage.
- A polypeptide is a single linear chain of many amino acids (any length), held together by amide bonds.
- A protein consists of one or more polypeptides (more than about 50 amino acids long).
- An oligopeptide consists of only a few amino acids (between two and twenty).
Number of amino acids
Peptides of defined length are named using IUPAC numerical multiplier prefixes.
- A monopeptide has one amino acid.
- A dipeptide has two amino acids.
- A tripeptide has three amino acids.
- A tetrapeptide has four amino acids.
- A pentapeptide has five amino acids.
- A hexapeptide has six amino acids.
- A heptapeptide has seven amino acids.
- An octapeptide has eight amino acids (e.g., angiotensin II).
- A nonapeptide has nine amino acids (e.g., oxytocin).
- A decapeptide has ten amino acids (e.g., gonadotropin-releasing hormone and angiotensin I).
Function
- A neuropeptide is a peptide that is active in association with neural tissue.
- A lipopeptide is a peptide that has a lipid connected to it, and pepducins are lipopeptides that interact with GPCRs.
- A peptide hormone is a peptide that acts as a hormone.
- A proteose is a mixture of peptides produced by the hydrolysis of proteins. The term is somewhat archaic.
- A peptidergic agent (or drug) is a chemical which functions to directly modulate the peptide systems in the body or brain. An example is opioidergics, which are neuropeptidergics.
See also
| Wikiquote has quotations related to: Peptide |
- Acetyl hexapeptide-3
- Beefy meaty peptide
- Collagen hybridizing peptide, a short peptide that can bind to denatured collagen in tissues
- Bis-peptide
- CLE peptide
- Epidermal growth factor
- Journal of Peptide Science
- Lactotripeptides
- Micropeptide
- Multifunctional peptide
- Neuropeptide
- Palmitoyl pentapeptide-4
- Pancreatic hormone
- peptide spectral library
- Peptide synthesis
- Peptidomimetics (such as peptoids and β-peptides) to peptides, but with different properties.
- Protein tag, describing addition of peptide sequences to enable protein isolation or detection
- Replikins
- Ribosome
- Translation (biology)
References
- Hamley, I W (September 2020). introduction to Peptide Science. Wiley. ISBN 9781119698173.
- Nelson, David L.; Cox, Michael M. (2005). Principles of Biochemistry (4th ed.). New York: W. H. Freeman. ISBN 0-7167-4339-6.
- Saladin, K (13 January 2011). Anatomy & physiology: the unity of form and function (6th ed.). McGraw-Hill. p. 67. ISBN 9780073378251.
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "proteins". doi:10.1351/goldbook.P04898
- "What are peptides". Zealand Pharma A/S. Archived from the original on 2019-04-29.
- Ardejani, Maziar S.; Orner, Brendan P. (2013-05-03). "Obey the Peptide Assembly Rules". Science. 340 (6132): 561–562. Bibcode:2013Sci...340..561A. doi:10.1126/science.1237708. ISSN 0036-8075. PMID 23641105. S2CID 206548864.
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "amino-acid residue in a polypeptide". doi:10.1351/goldbook.A00279
- Abba J. Kastin, ed. (2013). Handbook of Biologically Active Peptides (2nd ed.). ISBN 978-0-12-385095-9.
- Duquesne S, Destoumieux-Garzón D, Peduzzi J, Rebuffat S (August 2007). "Microcins, gene-encoded antibacterial peptides from enterobacteria". Natural Product Reports. 24 (4): 708–34. doi:10.1039/b516237h. PMID 17653356.
- Pons M, Feliz M, Antònia Molins M, Giralt E (May 1991). "Conformational analysis of bacitracin A, a naturally occurring lariat". Biopolymers. 31 (6): 605–12. doi:10.1002/bip.360310604. PMID 1932561. S2CID 10924338.
- Torres AM, Menz I, Alewood PF, et al. (July 2002). "D-Amino acid residue in the C-type natriuretic peptide from the venom of the mammal, Ornithorhynchus anatinus, the Australian platypus". FEBS Letters. 524 (1–3): 172–6. doi:10.1016/S0014-5793(02)03050-8. PMID 12135762. S2CID 3015474.
- Meister A, Anderson ME; Anderson (1983). "Glutathione". Annual Review of Biochemistry. 52 (1): 711–60. doi:10.1146/annurev.bi.52.070183.003431. PMID 6137189.
- Hahn M, Stachelhaus T; Stachelhaus (November 2004). "Selective interaction between nonribosomal peptide synthetases is facilitated by short communication-mediating domains". Proceedings of the National Academy of Sciences of the United States of America. 101 (44): 15585–90. Bibcode:2004PNAS..10115585H. doi:10.1073/pnas.0404932101. PMC 524835. PMID 15498872.
- Finking R, Marahiel MA; Marahiel (2004). "Biosynthesis of nonribosomal peptides1". Annual Review of Microbiology. 58 (1): 453–88. doi:10.1146/annurev.micro.58.030603.123615. PMID 15487945.
- Du L, Shen B; Shen (March 2001). "Biosynthesis of hybrid peptide-polyketide natural products". Current Opinion in Drug Discovery & Development. 4 (2): 215–28. PMID 11378961.
- "UsvPeptides- USVPeptides is a leading pharmaceutical company in India". USVPeptides.
- Payne JW (1976). "Peptides and micro-organisms". Advances in Microbial Physiology. 13: 55–113. doi:10.1016/S0065-2911(08)60038-7. ISBN 9780120277131. PMID 775944. Cite journal requires
|journal=(help) - Hummel J, Niemann M, Wienkoop S, Schulze W, Steinhauser D, Selbig J, Walther D, Weckwerth W (2007). "ProMEX: a mass spectral reference database for proteins and protein phosphorylation sites". BMC Bioinformatics. 8 (1): 216. doi:10.1186/1471-2105-8-216. PMC 1920535. PMID 17587460.
- Webster J, Oxley D; Oxley (2005). Peptide mass fingerprinting: protein identification using MALDI-TOF mass spectrometry. Methods in Molecular Biology. Methods in Molecular Biology™. 310. pp. 227–40. doi:10.1007/978-1-59259-948-6_16. ISBN 978-1-58829-399-2. PMID 16350956.
- Marquet P, Lachâtre G; Lachâtre (October 1999). "Liquid chromatography-mass spectrometry: potential in forensic and clinical toxicology". Journal of Chromatography B. 733 (1–2): 93–118. doi:10.1016/S0378-4347(99)00147-4. PMID 10572976.
- Tao, Kai; Makam, Pandeeswar; Aizen, Ruth; Gazit, Ehud (17 Nov 2017). "Self-assembling peptide semiconductors". Science. 358 (6365): eaam9756. doi:10.1126/science.aam9756. PMC 5712217. PMID 29146781.
- Tao, Kai; Levin, Aviad; Adler-Abramovich, Lihi; Gazit, Ehud (26 Apr 2016). "Fmoc-modified amino acids and short peptides: simple bio-inspired building blocks for the fabrication of functional materials". Chem. Soc. Rev. 45 (14): 3935–3953. doi:10.1039/C5CS00889A. PMID 27115033.
- Tao, Kai; Wang, Jiqian; Zhou, Peng; Wang, Chengdong; Xu, Hai; Zhao, Xiubo; Lu, Jian R. (February 10, 2011). "Self-Assembly of Short Aβ(16−22) Peptides: Effect of Terminal Capping and the Role of Electrostatic Interaction". Langmuir. 27 (6): 2723–2730. doi:10.1021/la1034273. PMID 21309606.
- Ian Hamley (2011). "Self-Assembly of Amphiphilic Peptides" (PDF). Soft Matter. 7 (9): 4122–4138. doi:10.1039/C0SM01218A.CS1 maint: uses authors parameter (link)
- Kai Tao, Guy Jacoby, Luba Burlaka, Roy Beck, Ehud Gazit (July 26, 2016). "Design of Controllable Bio-Inspired Chiroptic Self-Assemblies". Biomacromolecules. 17 (9): 2937–2945. doi:10.1021/acs.biomac.6b00752. PMID 27461453.CS1 maint: uses authors parameter (link)
- Kai Tao, Aviad Levin, Guy Jacoby, Roy Beck, Ehud Gazit (23 August 2016). "Entropic Phase Transitions with Stable Twisted Intermediates of Bio‐Inspired Self‐Assembly". Chem. Eur. J. 22 (43): 15237–15241. doi:10.1002/chem.201603882. PMID 27550381.CS1 maint: uses authors parameter (link)
- Donghui Jia, Kai Tao, Jiqian Wang, Chengdong Wang, Xiubo Zhao, Mohammed Yaseen, Hai Xu, Guohe Que, John R. P. Webster, Jian R. Lu (June 16, 2011). "Dynamic Adsorption and Structure of Interfacial Bilayers Adsorbed from Lipopeptide Surfactants at the Hydrophilic Silicon/Water Interface: Effect of the Headgroup Length". Langmuir. 27 (14): 8798–8809. doi:10.1021/la105129m. PMID 21675796.CS1 maint: uses authors parameter (link)
- Heitz, Marc; Javor, Sacha; Darbre, Tamis; Reymond, Jean-Louis (2019-08-21). "Stereoselective pH Responsive Peptide Dendrimers for siRNA Transfection". Bioconjugate Chemistry. 30 (8): 2165–2182. doi:10.1021/acs.bioconjchem.9b00403. ISSN 1043-1802. PMID 31398014.
- Boelsma E, Kloek J; Kloek (March 2009). "Lactotripeptides and antihypertensive effects: a critical review". The British Journal of Nutrition. 101 (6): 776–86. doi:10.1017/S0007114508137722. PMID 19061526.
- Xu JY, Qin LQ, Wang PY, Li W, Chang C (October 2008). "Effect of milk tripeptides on blood pressure: a meta-analysis of randomized controlled trials". Nutrition. 24 (10): 933–40. doi:10.1016/j.nut.2008.04.004. PMID 18562172.
- Pripp AH (2008). "Effect of peptides derived from food proteins on blood pressure: a meta-analysis of randomized controlled trials". Food & Nutrition Research. 52: 10.3402/fnr.v52i0.1641. doi:10.3402/fnr.v52i0.1641. PMC 2596738. PMID 19109662.
- Engberink MF, Schouten EG, Kok FJ, van Mierlo LA, Brouwer IA, Geleijnse JM (February 2008). "Lactotripeptides show no effect on human blood pressure: results from a double-blind randomized controlled trial". Hypertension. 51 (2): 399–405. doi:10.1161/HYPERTENSIONAHA.107.098988. PMID 18086944.
- Wu, Hongzhong; Ren, Chunyan; Yang, Fang; Qin, Yufeng; Zhang, Yuanxing; Liu, Jianwen (April 2016). "Extraction and identification of collagen-derived peptides with hematopoietic activity from Colla Corii Asini". Journal of Ethnopharmacology. 182: 129–136. doi:10.1016/j.jep.2016.02.019. PMID 26911525.