Transition metal carboxylate complex
Transition metal carboxylate complexes are coordination complexes with carboxylate (RCO2−) ligands. Reflecting the diversity of carboxylic acids, the inventory of metal carboxylates is large. Many are useful commercially, and many have attracted intense scholarly scrutiny. Carboxylates exhibit a variety of coordination modes, most common are κ1- (O-monodentate), κ2 (O,O-bidentate), and bridging.
-acetate-tetrahydrate-3D-balls.png.webp)
Acetate and related monocarboxylates
- Structures of Selected Metal Acetates
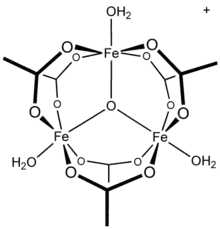 Basic ferric acetate
Basic ferric acetate2.png.webp) Silver acetate
Silver acetate
4.svg.png.webp) Molybdenum(II) acetate, illustrating the Mo-Mo quadruple bond.[1]
Molybdenum(II) acetate, illustrating the Mo-Mo quadruple bond.[1]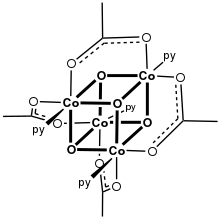 [CoO(acetate)]4, the "Das cubane"
[CoO(acetate)]4, the "Das cubane"
For simple carboxylates, the acetate complexes are illustrative. Most transition metal acetates are mixed ligand complexes. One common example is hydrated nickel acetate, Ni(O2CCH3)2(H2O)4, which features intramolecular hydrogen-bonding between the uncoordinated oxygens and the protons of aquo ligands. Stoichiometrically simple complexes are often multimetallic. One family are the basic metal acetates, of the stoichiometry [M3O(OAc)6(H2O)3]n+.[2]
Anhydrous or nearly anhydrous acetates often are [[coordination polymers. Some however adopt the Chinese lantern structure. Well studied examples include the dimetal tetraacetates (M2(OAc)4) where M = Cu(II), Rh(II), Cr(II), and Mo(II). Platinum diacetate and palladium diacetate feature Pt4 and Pd3 cores, further illustrating the tendency of acetate ligands to stabilize multimetallic structures.
2(H2O).svg.png.webp)
Reactions and applications
Metal acetates are common catalysts or precatalysts. Particularly useful are the lipophlic complexes of ethylhexanoates.
Di- and polycarboxylates
Oxalate complexes
- Structures of Some Metal Oxalate Complexes
5_tetraanion_(POWVOM).png.webp) [Cr2(oxalate)5]4-.
[Cr2(oxalate)5]4-.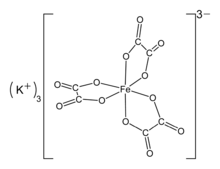 Potassium ferrioxalate (K
Potassium ferrioxalate (K
3[Fe(C
2O
4)
3]·3H2O)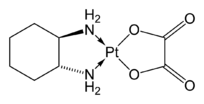 Oxaliplatin, an anticancer drug
Oxaliplatin, an anticancer drug4_tetraanion.svg.png.webp) [Zr(oxalate)4]4-
[Zr(oxalate)4]4-
Oxalate (C2O42-) is a distinctive dicarboxylate. As a small, symmetrical dinegative ion, oxalate commonly forms five-membered MO2C2 chelate rings. Mixed ligand complexes are known, e.g., [Co(C2O4)(NH3)4]κ+.[4]
Homoleptic oxalato complexes are common, e.g., those with the formula [M(κ2-C2O4)3]n-: M = V(III), Mn(III),[5] Cr(III), Tc(IV), Fe(III), Ru(III), Co(III), Rh(III), Ir(III). These anions are chiral (D3 symmetry), and some have been resolved into their component enantiomers. Some early metals form tetrakis complexes of the type [M(κ2-C2O4)4]3- M = Nb(V),[6] Zr(IV),[7] Hf(IV),[8] Ta(V),[9] Oxalate is often a bridging ligand forming bi- and polynuclear complexes with (κ2,κ'2-C2O4)M2 cores. Illustrative binuclear complexes are [M2(C2O4)5]2- M = Fe(II)[10] and Cr(III)[11]
Metal oxalate complexes are photoactive, degrading with loss of carbon dioxide. This reaction is the basis of the technique called actinometry. UV-irradiation of Pt(C2O4)(PPh3)2 gives derivatives of Pt0(PPh3)2.
Benzenedi- and tricarboxylates
Metal organic frameworks, porous, three-dimensional coordination polymers, are often derived from metal carboxylate clusters. These clusters, called secondary bonding units (SBU's), are often linked by the conjugate bases of benzenedi- and tricarboxylic acids.[12]
Aminopolycarboxylates
- Selected Complexes of Carboxylates
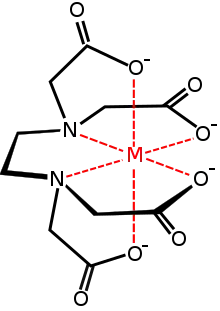 metal complex with the EDTA anion
metal complex with the EDTA anion2_anion.svg.png.webp) Ferric bis(iminodiacetate)
Ferric bis(iminodiacetate)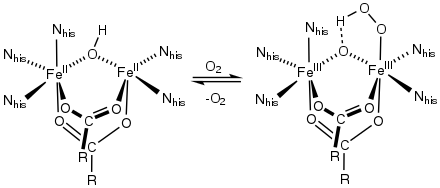 Active site of hemerythrin, an O2-carrying iron-carboxylate
Active site of hemerythrin, an O2-carrying iron-carboxylate
A commercially important family of metal carboxylates are derived from aminopolycarboxylates, e.g., EDTA4-. Related to these synthetic chelating agents are the amino acids, which form large families of amino acid complexes. Two amino acids, glutamate and aspartate, have carboxylate side chains, which function as ligands for iron in nonheme iron proteins, such as hemerythrin.[13]
References
- Brignole, Alicia B.; Cotton, F. A. (1972). "Rhenium and Molybdenum Compounds Containing Quadruple Bonds". Inorganic Syntheses. 13: 81–89. doi:10.1002/9780470132449.ch15.
- Catterick, Janet; Thornton, Peter (1977). Structures and Physical Properties of Polynuclear Carboxylates. Advances in Inorganic Chemistry and Radiochemistry. 20. pp. 291–362. doi:10.1016/S0065-2792(08)60041-2. ISBN 9780120236206.
- Zhang, Gao; Lin, Jian; Guo, Dong-Wei; Yao, Shi-Yan; Tian, Yun-Qi (2010). "Infinite Coordination Polymers of One- and Two-dimensional Cobalt Acetates". Zeitschrift für Anorganische und Allgemeine Chemie. 636 (7): 1401–1404. doi:10.1002/zaac.200900457.
- Bernal, Ivan; Cetrullo, James (1990). "The phenomenon of conglomerate crystallization. XIX. Clavic dissymmetry in coordination compounds. XVII". Structural Chemistry. 1 (2–3): 235–243. doi:10.1007/BF00674267. S2CID 96021604.
- Lis, T.; Matuszewski, J. (1980). "Structure of potassium tris(oxalato)manganate(III) trihydrate". Acta Crystallographica Section B. 36 (8): 1938–1940. doi:10.1107/S0567740880007558.
- Cotton, F. A.; Diebold, Michael P.; Roth, W. J. (1987). "Variable Stereochemistry of the Eight-Coordinate Tetrakis(oxalato)niobate(IV), Nb(C2O4)44-". Inorganic Chemistry. 26 (17): 2889–2893. doi:10.1021/ic00264a035.
- Fu, Yun-Long; Ren, Jia-Lin; Xu, Zhi-Wei; Ng, Seik Weng (2005). "Bis(4,4′-bipyridinium) Tetrakis(oxalato-κ2O,O′)zirconate(IV)". Acta Crystallographica Section E. 61 (11): m2397–m2399. doi:10.1107/S1600536805033829.
- Tranqui, D.; Boyer, P.; Laugier, J.; Vulliet, P. (1977). "Structure cristalline du tétrakisoxalatohafniate de Potassium Pentahydraté [K4Hf(C2O4)4.5H2O]". Acta Crystallographica Section B. 33 (10): 3126–3133. doi:10.1107/S0567740877010395.
- Perić, Berislav; Brničević, Nevenka; Jurić, Marijana; Planinić, Pavica; Matković-Čalogović, Dubravka (2009). "[NH4][(CH3)2NH2]2[Ta(C2O4)4]·2H2O: The First (Oxalato)tantalate(V) Complex Structurally Characterized". Structural Chemistry. 20 (5): 933–941. doi:10.1007/s11224-009-9494-0. S2CID 96838371.
- Armentano, Donatella; De Munno, Giovanni; Lloret, Francesc; Julve, Miguel (2005). "Bis and tris(oxalato)ferrate(III) complexes as precursors of polynuclear compounds". CrystEngComm. 7 (7): 57. doi:10.1039/b417251e.
- Masters, Vanessa M.; Sharrad, Clint A.; Bernhardt, Paul V.; Gahan, Lawrence R.; Moubaraki, Boujemaa; Murray, Keith S. (1998). "Synthesis, Structure and Magnetism of the oxalato-bridged chromium(III) complex [NBun4]4[Cr2(ox)5]·2CHCl3". Journal of the Chemical Society, Dalton Transactions (3): 413–416. doi:10.1039/a705265k.
- Tranchemontagne, David J.; Mendoza-Cortés, José L.; o'Keeffe, Michael; Yaghi, Omar M. (2009). "Secondary Building Units, Nets and Bonding in the Chemistry of Metal–Organic Frameworks". Chemical Society Reviews. 38 (5): 1257. doi:10.1039/b817735j. PMID 19384437.
- Jasniewski, Andrew J.; Que, Lawrence (2018). "Dioxygen Activation by Nonheme Diiron Enzymes: Diverse Dioxygen Adducts, High-Valent Intermediates, and Related Model Complexes". Chemical Reviews. 118 (5): 2554–2592. doi:10.1021/acs.chemrev.7b00457. PMC 5920527. PMID 29400961.