Tucatinib
Tucatinib, sold under the brand name Tukysa, is a small molecule inhibitor of HER2 for the treatment of HER2-positive breast cancer.[3][4] It was developed by Array BioPharma and licensed to Cascadian Therapeutics (formerly Oncothyreon, subsequently part of Seattle Genetics).[5]
 | |
| Clinical data | |
|---|---|
| Trade names | Tukysa |
| Other names | ONT-380, ARRY-380 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620032 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
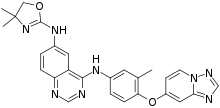
| Formula | C26H24N8O2 |
| Molar mass | 480.532 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Common side effects are diarrhea, palmar-plantar erythrodysesthesia (burning or tingling discomfort in the hands and feet), nausea, fatigue, hepatotoxicity (liver damage), vomiting, stomatitis (inflammation of the mouth and lips), decreased appetite, abdominal pain, headache, anemia and rash.[6][7] Pregnant or breastfeeding women should not take Tucatinib because it may cause harm to a developing fetus or newborn baby.[6]
Tucatinib was approved for medical use in the United States in April 2020,[6][7][8] and in Australia in August 2020.[1]
Medical uses
Tucatinib is a kinase inhibitor indicated in combination with trastuzumab and capecitabine for treatment of adults with advanced unresectable or metastatic HER2-positive breast cancer, including those with brain metastases, who have received one or more prior anti-HER2-based regimens in the metastatic setting.[2]
Clinical trials
Two early stage clinical trials have reported encouraging results, both of which had options to enroll subjects with central nervous system (CNS) metastases.[3][9][10][11][12] HER2CLIMB is a Phase 2 randomized, double-blinded, placebo-controlled study of tucatinib in combination with trastuzumab and capecitabine in patients with pretreated, unresectable locally advanced or metastatic HER2-positive breast cancer.[13]
History
In April 2020, the U.S. Food and Drug Administration (FDA) approved tucatinib in combination with chemotherapy (trastuzumab and capecitabine) for the treatment of adults with advanced forms of HER2-positive breast cancer that can't be removed with surgery, or has spread to other parts of the body, including the brain, and who have received one or more prior treatments.[6][7][14]
The FDA collaborated with the Australian Therapeutic Goods Administration (TGA), Health Canada, Health Sciences Authority (HSA, Singapore) and Swissmedic (SMC, Switzerland) on the review.[6] This was the first Project Orbis partnership between the FDA, HSA and Swissmedic.[6] As of 17 April 2020, the application is still under review at the other agencies.[6]
Tucatinib is a kinase inhibitor meaning it blocks a type of enzyme (kinase) and helps prevent the cancer cells from growing.[6] Tucatinib is approved for treatment after adults have taken one or more anti-HER2-based regimens in the metastatic setting.[6] The FDA approved tucatinib based on the results of the HER2CLIMB trial (NCT02614794) enrolling 612 subjects who had HER2-positive advanced unresectable or metastatic breast cancer and had prior treatment with trastuzumab, pertuzumab and ado-trastuzumab emtansine (T-DM1).[6][7] Subjects with previously treated and stable brain metastases, as well as those with previously treated and growing or untreated brain metastases, were eligible for the clinical trial, and 48% of enrolled subjects had brain metastases at the start of the trial.[6]
Subjects received either tucatinib 300 mg twice daily plus trastuzumab and capecitabine (tucatinib arm, n=410) or placebo plus trastuzumab and capecitabine (control arm, n=202).[7] The primary endpoint was progression-free survival (PFS), or the amount of time when there was no growth of the tumor, assessed by a blinded independent central review, evaluated in the initial 480 randomized patients.[6][7] The median PFS in subjects who received tucatinib, trastuzumab, and capecitabine was 7.8 months (95% CI: 7.5, 9.6) compared to 5.6 months (95% CI: 4.2, 7.1) in those subjects who received placebo, trastuzumab, and capecitabine (HR 0.54; 95% CI: 0.42, 0.71; p<0.00001).[6][7] Overall survival and PFS in subjects with brain metastases at baseline were key secondary endpoints.[6] The median overall survival in subjects who received tucatinib, trastuzumab, and capecitabine was 21.9 months (95% CI: 18.3, 31.0) compared to 17.4 months (95% CI: 13.6, 19.9) in subjects who received placebo, trastuzumab, and capecitabine (HR: 0.66; 95% CI: 0.50, 0.87; p=0.00480).[6][7] The median PFS in subjects with brain metastases at baseline who received tucatinib, trastuzumab and capecitabine was 7.6 months (95% CI: 6.2, 9.5) compared to 5.4 months (95% CI: 4.1, 5.7) in subjects who received placebo, trastuzumab and capecitabine (HR: 0.48; 0.34, 0.69; p<0.00001).[6][7]
The FDA granted the application for tucatinib priority review, breakthrough therapy, fast track, and orphan drug designations.[6][7][15] The FDA granted approval of Tukysa to Seattle Genetics, Inc.[6]
Society and culture
Legal status
Tucatinib was approved for medical use in the United States in April 2020.[6][7][8]
Tucatinib was approved for medical use in Australia in August 2020.[1]
On 10 December 2020, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Tukysa, intended for the treatment of HER2-positive locally advanced or metastatic breast cancer.[16] The applicant for this medicinal product is Seagen B.V.[16]
Names
Tucatinib is the International nonproprietary name.[17]
References
- "Tukysa". Therapeutic Goods Administration (TGA). 21 August 2020. Retrieved 22 September 2020.
- "Tukysa- tucatinib tablet". DailyMed. Retrieved 21 January 2021.
- "ONT-380 Active Against CNS Mets in HER2-Positive Breast Cancer". Cancer Network. 15 December 2015. Retrieved 17 April 2020.
- Martin M, López-Tarruella S (October 2018). "Emerging Therapeutic Options for HER2-Positive Breast Cancer". American Society of Clinical Oncology Educational Book. American Society of Clinical Oncology. Annual Meeting. 35 (36): e64–70. doi:10.1200/EDBK_159167. PMID 27249772.
- "Tucatinib" (PDF). Statement on a Nonproprietary Name Adopted by the USAN Council.
- "FDA Approves First New Drug Under International Collaboration, A Treatment Option for Patients with HER2-Positive Metastatic Breast Cancer". U.S. Food and Drug Administration (FDA) (Press release). 17 April 2020. Retrieved 17 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - "FDA approves tucatinib for patients with HER2-positive metastatic brea". U.S. Food and Drug Administration (FDA). 17 April 2020. Retrieved 20 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - "Drug Approval Package: Tukysa". U.S. Food and Drug Administration (FDA). 18 May 2020. Retrieved 21 January 2021.
- "Oncothyreon Inc. Announces Data For ONT-380 In HER2-Positive Breast Cancer Patients With And Without Brain Metastases At The San Antonio Breast Cancer Symposium". BioSpace (Press release). 9 December 2015. Retrieved 18 April 2020.
- "SABCS15: Promising phase 1 results lead to phase 2 for ONT-380 in HER2+ breast cancer". Colorado Cancer Blogs. Retrieved 10 June 2016.
- "A Study of Tucatinib (ONT-380) Combined With Capecitabine and/or Trastuzumab in Patients With HER2+ Metastatic Breast Cancer". ClinicalTrials.gov. 31 December 2013. Retrieved 18 April 2020.
- Borges VF, Ferrario C, Aucoin N, Falkson CI, Khan QJ, Krop IE, et al. "Efficacy results of a phase 1b study of ONT-380, a CNS-penetrant TKI, in combination with T-DM1 in HER2+ metastatic breast cancer (MBC), including patients (pts) with brain metastases". Journal of Clinical Oncology. 2016 ASCO Annual Meeting.
- "A Study of Tucatinib vs. Placebo in Combination With Capecitabine & Trastuzumab in Patients With Advanced HER2+ Breast Cancer (HER2CLIMB)". ClinicalTrials.gov. Retrieved 18 April 2020.
- "Tukysa: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 20 April 2020.
- "Tucatinib Orphan Drug Designation and Approval". U.S. Food and Drug Administration (FDA). 24 December 1999. Retrieved 20 April 2020.
- "Tukysa: Pending EC decision". European Medicines Agency (EMA). 10 December 2020. Retrieved 11 December 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- World Health Organization (2016). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 75". WHO Drug Information. 30 (1): 161. hdl:10665/331046.
External links
- "Tucatinib". Drug Information Portal. U.S. National Library of Medicine.
- "Tucatinib". National Cancer Institute.
- Clinical trial number NCT02614794 for "A Study of Tucatinib vs. Placebo in Combination With Capecitabine & Trastuzumab in Patients With Advanced HER2+ Breast Cancer (HER2CLIMB)" at ClinicalTrials.gov